Image: Adrian Lange on Unsplash
Biofilms are prolific and problematic. They cause large economic losses, from the failure of medical implants to enhanced erosion of pipes in our cities. However, scientists have devised a low-cost method to “clean up” biofilms, which could save society money, lives, and resources.
What are biofilms?
Biofilms are colonies of microorganisms, which can include algae, bacteria, and protozoa. These multicellular communities produce a glue-like substance to stick themselves — often in a matter of minutes — to a surface at a solid-liquid interface.
Bacterial biofilms are particularly challenging. The bacteria cover themselves with protective mucus and are more resistant to disinfectants and antibiotics.
“These have profound negative impact,” said Professor Martin Pumera, principal investigator for the study.
In medicine, biofilms are frequently found on medical implants, teeth, catheters, and other surfaces, increasing the risk of infection or failure of the implant. They can also corrode water pipelines and contaminate the water — affecting taste, odor, color, and safety.
“They cause about 100 billion USD annual loss just in USA,” said Pumera. Therefore, a robust method to effectively remove bacterial biofilms would be exceedingly valuable.
Biofilm-fighting microrobots
Micromotors — and their smaller counterparts, nanomotors — are synthetic objects that are autonomously self-propelled by using energy from their environment.
Motion is facilitated by asymmetric structures, such as microjet engines or helix swimmers. The symmetry of the molecule breaks when the appropriate stimulus, such as a chemical fuel, is applied. This produces a gradient (electrical, chemical concentration, surface tension, or temperature) or the formation of bubbles, which in turn propels the motors.
Few studies to date have considered using micromotors to deal with biofilms, and most existing micromotors are short-lived and expensive to create.

However, Pumera and colleagues synthesized micromotors made of zinc oxide doped with silver (ZnO:Ag) — both of which have antibacterial properties — using a relatively low-cost and environmentally benign method. The micromotors were stable, round-shaped stars (a rough ball covered with protruding microrods) that self-propel under UV light.
In laboratory tests, these micromotors killed more than 80% of the tested biofilms within 5 minutes. In particular, the bacteria Pseudomonas aeruginosa, which is often found in wounds and drinking water pipes, was killed with a very small amount of motors (1 μg mL−1) in a short time. In addition to causing cell death, the motors caused damage that hindered the formation of new biofilm.
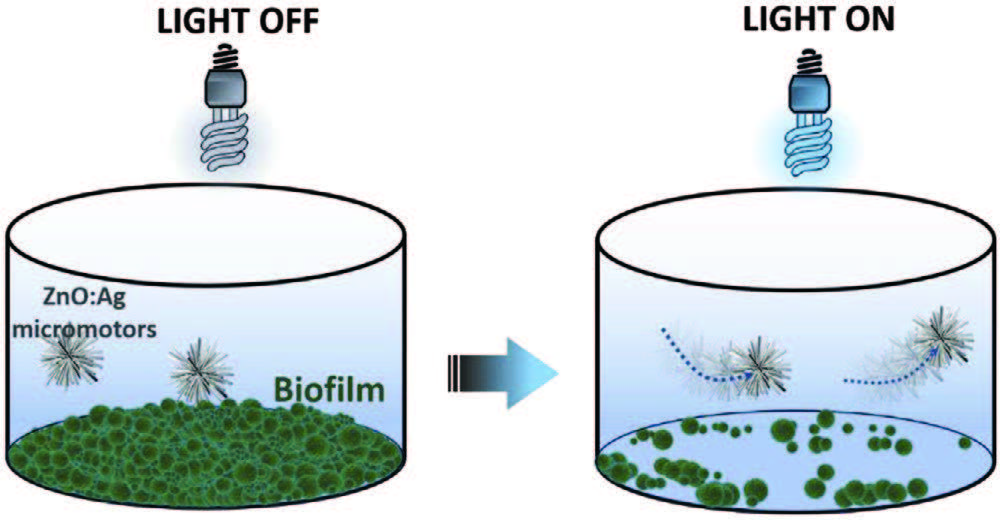
These results create opportunities for the broad application of micromotors in biofilm removal in healthcare, industry, and environmental fields.
“We are now focusing on medical application of micro and nanorobotics in removal biofilms in medicine,” said Pumera. “We are also working on collective behavior of these motors, so they can act together and in coordinated action attack the bacteria.”
Reference: M. Ussia, et al., Active Light‐Powered Antibiofilm ZnO Micromotors with Chemically Programmable Properties, Advanced Functional Materials (2021). DOI: 10.1002/adfm.202101178