Contraception has become essential in today’s society—as birth control, for family planning, to promote economic growth. The benefits are plenty, from preventing unwanted pregnancy and reducing the need for abortion, to improving the health and well-being of women.
Current methods are largely hormone based, administered either orally, transdermally, intravaginally, or intrauterinely, with the ‘pill’ being the most common. Alternatively, a surprising number of women opt for female sterilization, followed by the use of male condoms or the IUD. There are currently no hormone-based male contraceptive methods available, limiting the choice to male condoms or sterilization (vasectomy). However, when it comes to hormone agents, the benefits don’t come without cost, namely the effects on women’s health.
Clearly, there is a great need for new developments in this field, including both female contraceptives with fewer side effects and the introduction of male contraceptives that are on par.
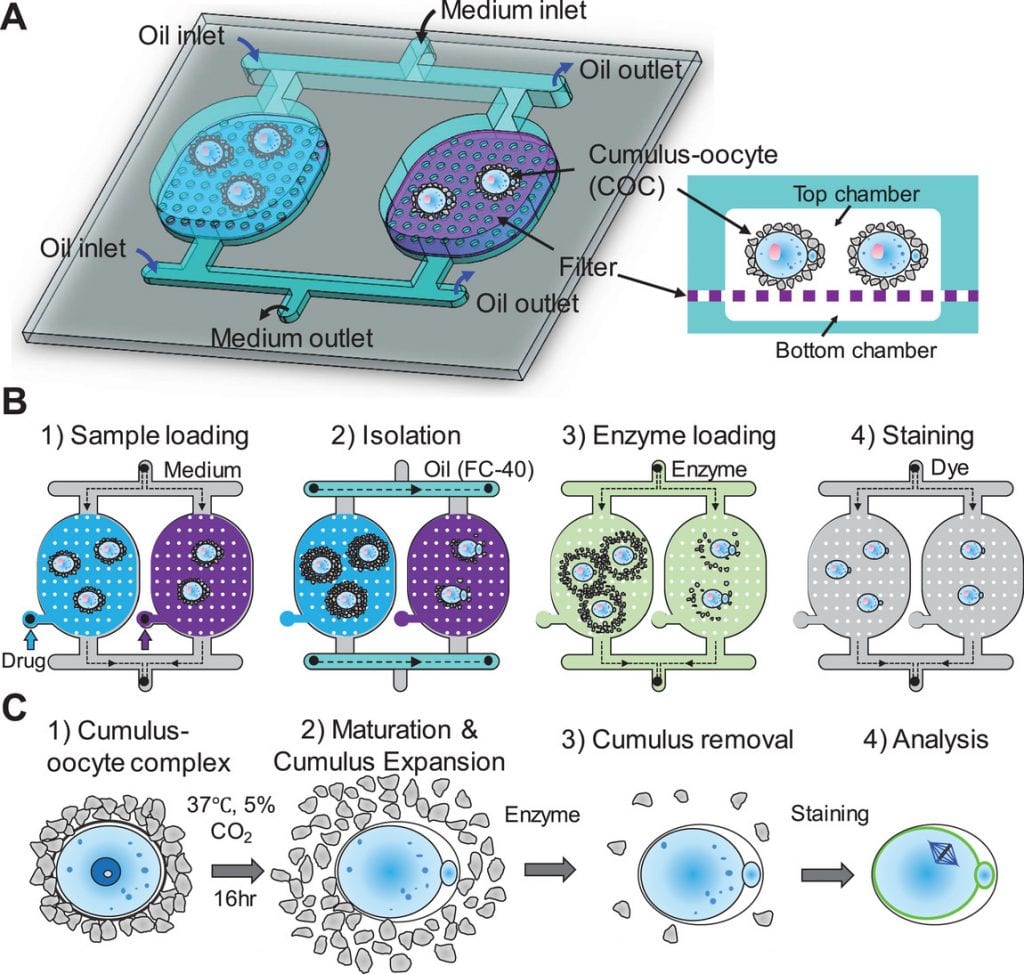
To this end, Hui Li and co-workers at Pennsylvania State University have developed a method for analyzing and screening nonhormonal contraceptive agents. In particular, their device looks at the cumulus–oocyte complex (COC), which facilitates transfer of the egg to the oviduct. They suggest that targeting both cumulus expansion and oocyte maturation may enable safer and more easy-to-use contraception or combination regimes.
In the recently published article in Small, a multiwell microfluidic device is developed (see schematic) that is capable of screening multiple drugs in parallel. Additionally, it facilitates COC incubation, medium exchange, enzyme loading, removal of cumulus cells, and fluorescence staining in each well, and was shown to be feasible for screening nonhormonal contraceptives by studying the effects of three nonhormonal agents on COC development.
The 3D printed device is a simple and scalable platform, which could be a promising alternative to current manual screening methods. With future aspirations to incorporate automated imaging and analysis, or other processes such as on-chip fertilization, this research holds the potential to be used for evaluation of not only contraceptive agents on the COC, but could be expanded to study various stages of reproduction.