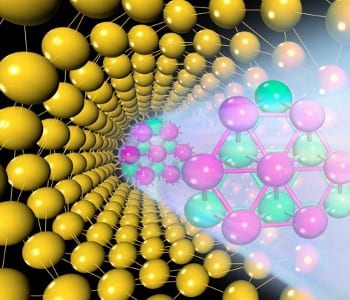
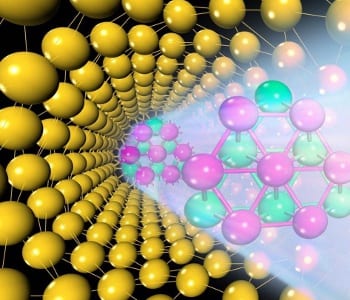
 A new 1D gold nanowire is proposed for novel nanoelectronics by researchers in China. The design of the new structure was achieved by carefully considering the stability of 0D, 1D, and 2D gold nanostructures. The nanostructure consists of a core of hexagonal-close-packed gold atoms, wrapped by a curved shell with a (111) face-centered-cubic (FCC) surface structure. In theoretical calculations, the new nanowire is found to be more stable than other types of gold nanowires, including structures that can be experimentally synthesized. The researchers suggest that the proposed structure could be achieved by thinning gold wires down to 1.5 nm in a protective atmosphere, followed by thermal annealing.
A new 1D gold nanowire is proposed for novel nanoelectronics by researchers in China. The design of the new structure was achieved by carefully considering the stability of 0D, 1D, and 2D gold nanostructures. The nanostructure consists of a core of hexagonal-close-packed gold atoms, wrapped by a curved shell with a (111) face-centered-cubic (FCC) surface structure. In theoretical calculations, the new nanowire is found to be more stable than other types of gold nanowires, including structures that can be experimentally synthesized. The researchers suggest that the proposed structure could be achieved by thinning gold wires down to 1.5 nm in a protective atmosphere, followed by thermal annealing.
The smooth surface structure leads to low chemical activity on the nanowire, making it non-reactive and robust even when exposed to air. Additionally, in contrast to most nanowire structures, the high work function of the surface will ensure that the current in the nanowire is stable when used in electronic devices. The large variation in conductance around the Fermi level also enables the conductance to be tuned with bias voltage.
Since the discovery of graphene and other low-dimensional carbon materials, analogue structures for transition metals have also been sought. Gold has received particular attention, with various nanoclusters, nanocages, and nanowires existing stably. However, 2D structures are elusive. Through spin-polarized density functional calculations, the researchers determined the origin of the instability, and revealed the differences between carbon and transition metals that allow carbon to exist stably in the various nanostructures. In carbon, 2D graphene structures are bonded with sp2 hybridized bonds, whereas, for the 3D diamond structure, the bonds are sp3 hybridized. Bonds are formed with electrons in the 2s and 2p orbitals, with 2 and 3 p states participating respectively in the sp2 and sp3 hybrid bonds. Transition between the sp2 and sp3 bonds requires overcoming a large energy barrier, and hence both 2D and 3D forms are stable. In gold and other transition metals, d and s orbitals are involved in bonding. In bulk FCC metals, the bonds are sd5 hybrid bonds—s states mixed with five d states. For 2D structures, the researchers found that the bonds have a predominantly sd4 hybrid character, with only four d states involved. Because there is no significant energy difference between the sd5 and sd4 states, transforming from a 2D structure to a 3D structure at certain atom numbers in energetically favorable, and thus the 2D structure is unstable. For silver, platinum, and gold, the threshold atom number for the 2D-to-3D transition is 7, 10, and 14 respectively. Other transition metals are not expected to form similar stable nanowire structures.