In nanomedicine, off-site toxicity, low accumulation of drug in the target tissue, and poor response from patients remain critical shortcomings. Bringing our most up-to-date understanding of ligand-target relationships into the design of targeted nano-carriers, however, may finally allow nanomedicines to flourish in the clinic.
Ideal drug targets would be highly expressed on diseased cells with only low expression on healthy cells. However, very few receptors fit this requirement. While targeted nanocarriers are emerging as a promising tool to enhance the ‘druggability’ of less-than ideal targets, clinical translation is hindered due to a lack of appreciation for the complexity of ligand-receptor relationships.
In a review published in WIREs Nanomedicine and Nanobiotechnology, “Precision engineering of targeted nanocarriers,” Michael Deci, Maixian Liu, Quoc Thai Dinh, and Juliane Nguyen from the University at Buffalo highlight how nanocarriers can be precision-engineered to capitalize on what is known about receptor expression patterns, receptor clustering, and ultimately how interactions between a nanocarrier and target can be manipulated to achieve the most effective downstream signaling events.
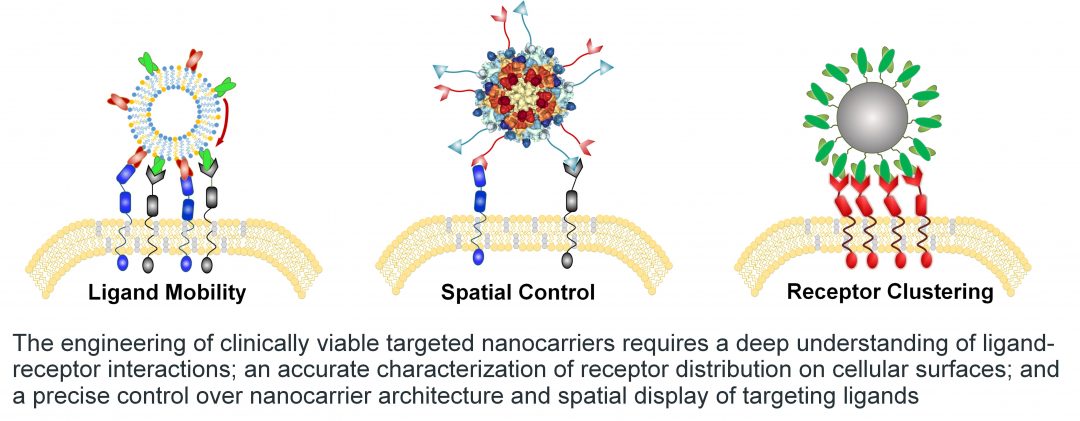
The authors discuss topics such as membrane fluidity and spatial control of ligand display, patterning of receptors, and importantly, how clustering of receptors can serve to amplify downstream signaling events. The review also notes that while spatiotemporal changes in receptor expression upon external cues and disease progression is reported, these changes are often disregarded and not considered in the design of nanocarriers. In many instances, targeted nanocarriers are engineered assuming a ‘static’ target, even though it is clear that throughout the course of many diseases this simply is not the case.
Since conception, the relationship between targeted nanocarriers and receptors have been oversimplified. To achieve robust engagement of a ligand with its target necessitates a better understanding of factors controlling receptor engagement is of utmost importance. Membrane fluidity, multivalency, and spatial control are highlighted as points of manipulation for designing nanocarriers that effectively treat disease. Understanding how modulation of these parameters can equip nanocarriers with spatiotemporal responsiveness and enhanced biological activity will be essential to make targeted nanomedicine a clinical reality.
Kindly contributed by the Authors.