Being able to do one job well just isn’t good enough any more.
At least twenty years ago, the properties of nanoparticles useful for medical therapy and diagnostics were already being seriously explored, with a view to their clinical application. Now, multifunctional particles – those that can perform many jobs at once or sequentially, as needed – are the more important avenue of research.
This multifunctionality has a few obvious practical benefits – it has the potential to reduce the strain on important and sometimes scarce resources by making them work harder, and it could render medical testing and therapy less invasive or difficult for both patient and clinician, requiring only one dose of something to allow multiple different procedures.
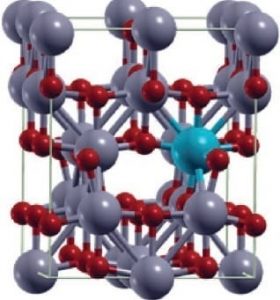
Titania doped with gadolinium shows promise for combining medical imaging and cancer therapy.
A nice example of such multifunctionality comes from a large collaboration of researchers working across Germany, Sweden, Slovenia, and Russia, who take nanobeads of TiO2 and dope them with gadolinium.
TiO2 (titania) is most commonly used as a white pigment in paints and foods, and is found in sunscreen as a protective component due to its high refractive index and resistance to UV damage. The properties that make it useful in these applications also make it interesting for photodynamic therapy (PDT), which endeavors to kill cancerous cells by exploiting their high sensitivity to nearby increases in reactive oxygen species (ROS). In this context, TiO2 can act as a very efficient photosensitizer, generating a large amount of ROS inside the body in response to externally applied radiation.
What needs to be avoided, however, is increased ROS generation all over the body, leading to the destruction of normal, healthy cells. Unfortunately, TiO2 does not have any properties useful for the types of imaging that would allow it to be tracked and monitored inside the body, and its modification with appropriate surface-capping ligands reduces its ROS generation efficiency.
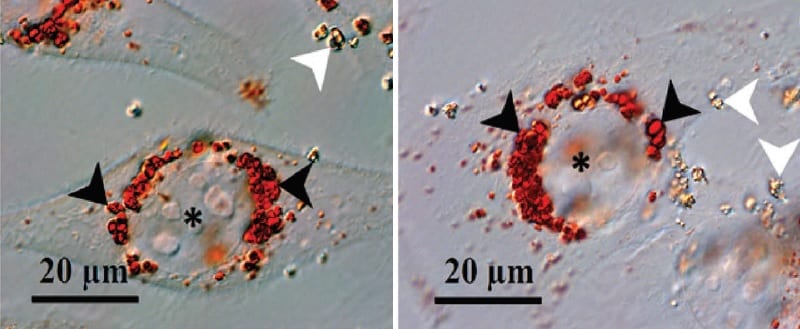
Gadolinium:titania particles inside cells: acid organelles are stained with a red dye, and are enlarged in the presence of the particles (black arrows). Asterisk indicates the cell nucleus.
Therefore, this research incorporates gadolinium inside 100-nm-sized TiO2 nanoparticles, leaving the surface clean and free for ROS generation. Nanoparticle sizes of 100 nm and slightly above present a high reactive surface area, but have been demonstrated to avoid cytotoxicity due to nanoscale surface effects, and gadolinium is already used as a contrast agent for magnetic resonance imaging (MRI).
The doped nanoparticles allowed simultaneous imaging and PDT, providing an impressive multifunctionality without the need to compromise the advantages of either component: the doped TiO2 particles showed a better imaging contrast and even better ROS generation compared to the undoped particles.
For more details and information on the optimum doping level of gadolinium, please see the Full Paper in Small.
If you’re doing research into PDT or other cancer therapies, perhaps you should check out these articles:
A Potent Magnetic Fluid Hyperthermia Agent for Cancer Treatment
Multimodal Agent for Cancer Diagnostics and Therapeutics
Featured Image Credit: Nuclear Regulatory Commission via Flickr, CC-BY 2.0