With the aim of modeling the detailed structure and functions of living native tissues, tissue engineering strategies have been developed to create three dimensional (3D) platforms that mimic the composition and spatial distribution of their cellular components. The developed platforms can be implanted, opening new therapeutic options for diseases and injuries that currently have no effective treatment options. However, the construction of complex biological structures, such as the spinal cord, requires both the control of biomaterial distribution and spatially accurate placement of multiple cell types, which remain significant challenges.
Recently published in Advanced Functional Materials, a multidisciplinary team of researchers from the University of Minnesota has developed a method to model complex tissue architecture with an advanced, custom-built, one-pot 3D cell printing process.
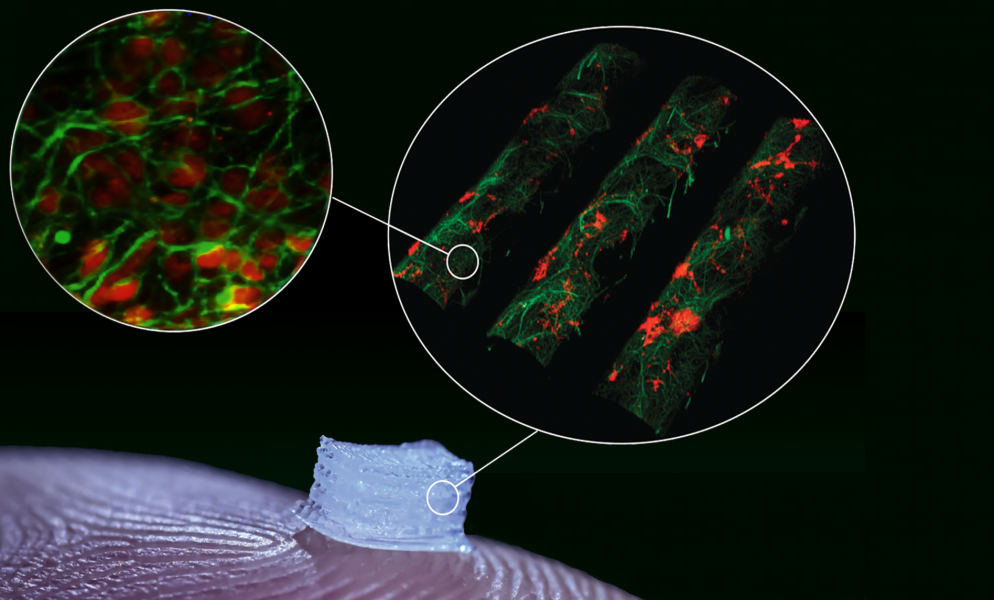
In their “living” spinal cord model, a neuro-compatible multichannel scaffold was printed in which different types of stem-cell derived neural progenitor cells were precisely placed in designated locations. The printed progenitor cells could then differentiate and extend axons throughout the scaffold channels, generating the functional activity of neuronal networks, which is a critical foundation for therapeutic devices.
The successful printing of different types of progenitor cells directly onto the scaffold enables a multicellular neural tissue engineering approach, which allows for the ability to control the position, growth, and differentiation of transplanted cells and may be beneficial in rebuilding connections across areas of tissue damage.
The next challenge is to transplant these scaffolds in an in vivo model injury to assess their effect on tissue function as well as the survival and fate of the cells. Prof. Michael C. McAlpine, co-senior author of the study, hopes “this living platform could ultimately be used to prepare novel biomimetic scaffolds modeling complex tissue architectures, in order to develop a clinical implant for treating neurological diseases, including spinal cord injury.”