The current seasonal influenza vaccine provides protection specifically against three or four strains of influenza, but frequently the strains selected for the seasonal influenza vaccine are not the strains we encounter during flu epidemics.
Therefore, the seasonal flu vaccines do not provide significant protection against flu pandemics that are, according to the World Health Organization, a matter of “not if, but when”.
Flu Pandemics
We’re all familiar with the worries of flu pandemics. All of us reading this will remember the flu pandemic of 2009–2010, more commonly referred to as “swine flu” due to it originating in pigs. This particular strain of the H1N1 virus was believed to have infected between 10 and 20% of the global population, of whom several hundred thousand died as a result. But a far more deadlier pandemic from H1N1 occurred in 1918–1919.
What came to be known as the Spanish Flu broke out in the closing months of the First World War, and quickly spread around the world, claiming the lives of between 50 and 100 million people. Indeed, some believe it was conditions of the war itself—close quarters of soldiers, the mass movement of troops, already-compromized immune systems of soldiers in the trenches—that caused the virus to spread so far and so quickly, as well as possibly accelerating its mutation rate and potency.
The Spanish Flu was a global trauma felt by the vast majority of people alive at the time, and is a somber reminder of just how unpredictable and deadly flu outbreaks can be.

The swine flu pandemic ten years ago therefore justifiably caused great concern from public and health officials alike. It’s because of deadly outbreaks like these, and the big question mark as to when the next one will be, that scientists are working hard to find a “universal” flu vaccine—one that protects us against the deadly, as well as the more common, seasonal strains.
A Promising New Vaccine
Dr. Baozhong Wang’s team at the Institute for Biomedical Sciences at Georgia State University is developing a vaccine that they hope will protect against all strains of influenza by using parts of influenza proteins that are common to most influenza strains.
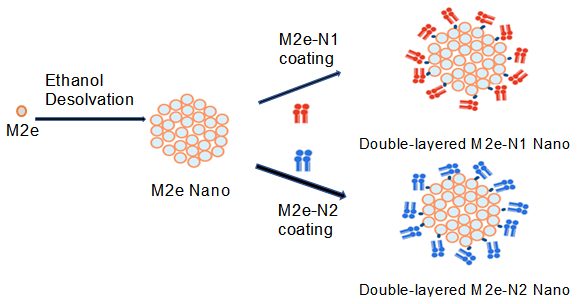
We orchestrate these shared structures into multi-layered protein nanoparticles as flu vaccine candidates to induce immune responses conferring a “universal” protection against different flu viruses. In a study recently published in Advanced Healthcare Materials, we generated double-layered protein nanoparticles with influenza Matrix protein 2 ectodomain and neuraminidase.
During immunization with the seasonal influenza vaccine, the human immune system generates responses against the head region of the surface protein hemagglutinin. This type of immune response provides very narrow protection because the head region of hemagglutinin is highly variable between strains.
“Using a preliminary mouse model, our vaccines are able to provide broad protection against many flu strains by using conserved influenza peptides,” says Gonzalez. “Some of the conserved peptides we have been experimenting with are the hemagglutinin stem, neuraminidase, and the Matrix 2 protein ectodomain.”
There has been research in the past with subunits of influenza but these vaccines weren’t very effective. The soluble protein in these subunit vaccines is quickly “swept away” and few helpful immune responses are generated.
Uniquely, we have been able to improve the immunogenicity of the peptides in our vaccines by formulating them into nanoparticles. The nanoparticles protect the peptides from degradation and interact with immunity-generating, antigen-presenting cells in an adjuvant-like fashion.
Whilst the next level would be to see whether such nanoparticle-based vaccines work in humans, the results from the mouse models are an encouraging step toward this goal.
“Currently, we are experimenting with new combinations of peptides, protein adjuvants, and dissolvable microneedle patches as a way to deliver our nanoparticles. A successful universal influenza vaccine will protect everyone from future flu epidemics, saving lives and money.”
Kindly contributed by Gilbert X. Gonzalez, Institute for Biomedical Sciences, Georgia State University.