Hydrogels are water-swollen ne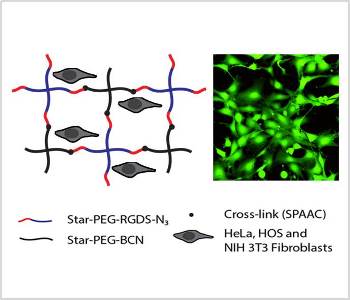 tworks, formed by cross-links between polymeric constituents. Due to their high water content and tunable mechanical properties, hydrogels are highly promising materials for tissue engineering and controlled drug delivery applications. With tissue engineering as an application in mind, gels need to be prepared with mechanical properties that mimic the stiffness of mammalian organs. In this respect, poly(ethylene)glycol (PEG) hydrogels are very promising materials.
tworks, formed by cross-links between polymeric constituents. Due to their high water content and tunable mechanical properties, hydrogels are highly promising materials for tissue engineering and controlled drug delivery applications. With tissue engineering as an application in mind, gels need to be prepared with mechanical properties that mimic the stiffness of mammalian organs. In this respect, poly(ethylene)glycol (PEG) hydrogels are very promising materials.
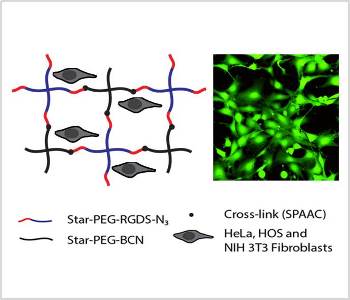
The cross-links can be either of a physical or chemical nature. Chemical cross-links are often introduced by click reactions which are high-yielding, fast and bio-orthogonal. One particularly useful example is the copper-free click reaction between a ring-strained alkyne and an azide (SPAAC). This reaction can successfully be performed without the addition cytotoxic copper ions which are found to be extremely difficult to be completely removed. Bio-orthogonality is required if biomacromolecules like peptides or proteins should be attached to or included into a hydrogel.
Recently, researchers from Radboud University (Nijmegen, NL) prepared soft biocompatible PEG hydrogels with tunable mechanical properties and RGDS-content using the copper free-click reaction. Rheological analysis showed that the resulting hydrogels display mechanical properties comparable to that of soft tissues. The stiffness of the gels could be tuned by changing the polymer density and the gels were modified with cell adhesion motif RGDS to ensure biocompatibility. Cells were shown to remain viable and showed a nicely spread morphology on the RGDS-containing hydrogels. The cross-linking method is biocompatible and yields good substrates for cellular adhesion.
Cellular interactions with gel surfaces were studied using hydrogels with different stiffness. Three cell types were tested. Whereas bone marrow derived HOS cells were found to be able to spread on all RGDS-containing gels, independent of their stiffness, cells derived from softer tissues as HeLa and NIH 3T3 fibroblasts showed decreasing cellular adherence with increasing gel stiffness. Hence, the observed interaction of cells with these particular hydrogel surfaces showed that gel stiffness influences cellular adhesion.