The use of nanoparticles in the formulation of vaccines has advantages, for example the possibility to target the lymphoid organs, and the enhancement of the function for loaded therapeutics. Porous silicon (PSi) provides a versatile biocompatible simple or multistage platform for various therapeutic applications. Biomimetic cell-derived materials such as vesicles derived from the cell membrane of leukocytes or red blood cells encapsulate polymeric particles in multistage vectors, and have been found to prolong the payload circulation time, reduce the interaction with macrophages and the nanoparticle uptake in the liver. Indeed, vesicles derived from cancer cells can be successfully employed in cancer immunotherapy. Combining the two approaches could have advantages.
Flavia Fontana, Hélder Santos and colleagues from the University of Helsinki and the University of Turku, Finland, and Harvard University, USA, have developed two multistage nanovaccines constructed of two biomaterials and either a biological cancer cell membrane (CCM) or a model antigen. The systems show immune-stimulant properties in human cells, promoting the expression of costimulatory signals and the secretion of pro-inflammatory cytokines.
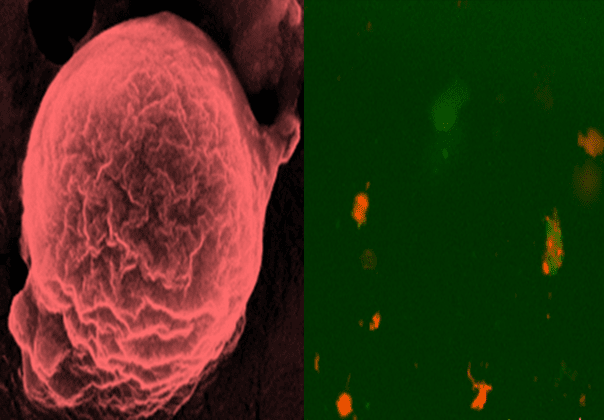
A glass‐capillary microfluidics nanoprecipitation technique was used to produce the initial two layers of a nanovaccine. TOPSi nanoparticles were encapsulated into AcDEX or spermine‐modified AcDEX (SpAcDEX) polymeric particles (see Figure). The particles coated with AcDEX were then co‐extruded together with vesicles derived from CCM to obtain the final core–shell system (TOPSi@AcDEX@CCM). TOPSi@SpAcDEX particles were functionalized with a model antigen, Trp2, to provide the second system (TOPSi@SpAcDEX‐Trp2).
These materials offer opportunities in the field of personalized medicine, with the possibility of functionalizing the cancer vaccine with antigens derived from the cancer cells of each individual patient.