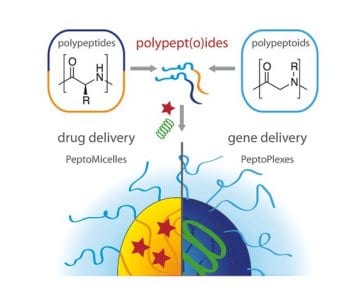
Polypept(o)ides are defined as polypeptide-block-polypeptoid copolymers. This hybrid material furthermore combines intrinsic multifunctionality, enzymatic degradability, distinct secondary structure formation, and stimuli-responsivene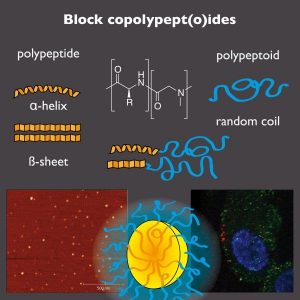 ss of polypeptides with the stealth-like properties of polypeptoids, e.g. polysarcosine.
ss of polypeptides with the stealth-like properties of polypeptoids, e.g. polysarcosine.
Polysarcosine (PSar) has already displayed its great potential as a substitute for PEG in biomedical applications. Like PEG, PSar is hydrophilic, non-ionic and exclusively a weak hydrogen bond acceptor. Therefore, it is not much of a surprise that PSar has been shown to be protein-resistant. It can also reduce immunogenicity of peptides and proteins and elongate their circulation times. Additionally, sarcosine is a cheap non-chiral endogeneous amino acid, which can be easily transferred into the corresponding NCA and polymerized under living conditions. A multitude of functional end groups can be introduced, which are often impossible to incorporate into polymers synthesized by anionic polymerization. Additionally, polysarcosine can be employed as macroinitiator allowing for convenient and scalable synthesis of block copolypept(o)ides by sequential ring opening polymerization of the corresponding α-amino acid N-carboxy-anhydrides (NCAs). Since exclusively endogeneous amino acids are employed, polypept(o)ides can be used for the synthesis of polymers and functional nanoparticles bearing the great promise of being stable in blood circulation but being completely metabolized into non-toxic components, amino acids, by proteases inside cells.
Surprisingly, publications on polypept(o)ides are so far highly limited. In a recent article Matthias Barz and Kristina Klinker provide a first overview concerning the work on polypept(o)ides, summarizing synthetic approaches, self-assembly behavior in solution, and initial applications. Moreover, future applications and research directions for these novel materials are presented.