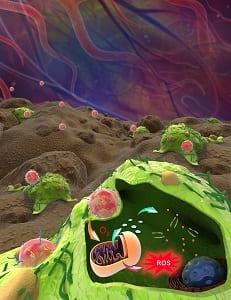
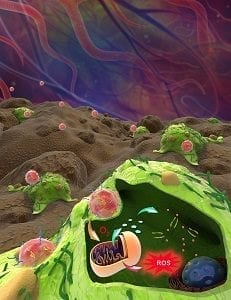
 A newly developed nanocarrier shows excellent oxygen-carrying and ATP-responsive drug release properties, which makes it a potent agent for cancer therapy. The oxygen nanocarrier (A/D-ONC) combines a polymeric core entrapping hemoglobin and a cationic lipid shell absorbing doxorubicin-intercalated DNA.
A newly developed nanocarrier shows excellent oxygen-carrying and ATP-responsive drug release properties, which makes it a potent agent for cancer therapy. The oxygen nanocarrier (A/D-ONC) combines a polymeric core entrapping hemoglobin and a cationic lipid shell absorbing doxorubicin-intercalated DNA.
Hemoglobin is a natural oxygen-transport protein in red blood cells. Because of hemoglobin encapsulation this nanocarrier-based drug delivery system can form artificial ‘red blood cells’ with a good oxygen-carrying capacity. By donating oxygen to cells, oxygenated A/D-ONC can greatly influence the mitochondrial metabolism of cancer cells.
However intracellular ATP – which promotes cell growth – and reactive oxygen species (ROS) – which damages cancer cells – are both generated by mitochondria. To achieve ideal anticancer efficacy, it is necessary to utilize both for cancer therapy. By applying ATP-responsive DNA to intercalate chemotherapy drug doxorubicin, A/D-ONC can realize ATP-triggered intracellular drug release, and hence successfully convert growth-beneficial ATP to a trigger of chemotherapy. After cell uptake, the oxygenated A/D-ONC donates excessive oxygen to cancer cells and kills them in two ways: 1. by increasing intracellular ATP content to promote ATP-responsive drug release for cancer chemotherapy and, 2. by synchronously increasing intracellular ROS amount to amplify its lethality to cancer cells.
By turning the growth-beneficial ATP to chemotherapy activator and enhancing ROS lethality, A/D-ONC offers a novel drug delivery approach for combined cancer therapy.