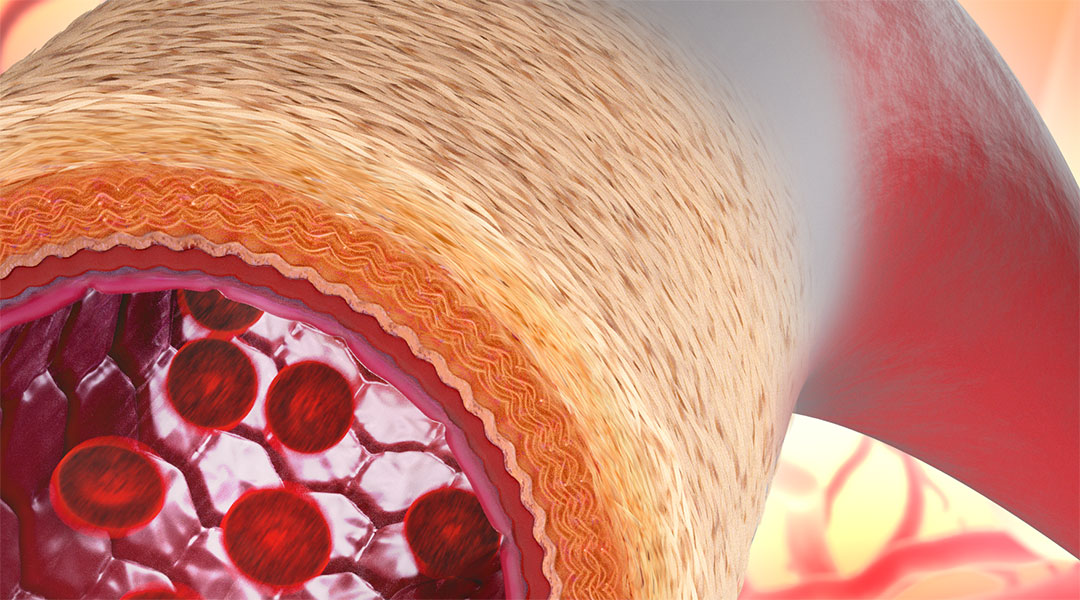
Some 60,000 miles of biological pipework weave a dense blood transport network through our body. Inevitably, leaks and blockages can occur, and the fix is a littler harder than replacing a pipe in your bathroom.
The consequences of compromised blood vessels can range from a small bruise to myocardial infarction — a block of blood flow to the heart that is the leading cause of death worldwide. Replacements can be life changing or lifesaving, but blood vessels are a feat of engineering and biology not easily replicated.
Swapping in vessels from elsewhere in the body isn’t always possible, and artificial approximations have struggled to match the true complexity of natural vessels. A new approach, detailed in Advanced Materials, has created a manufactured vessel that mimics our vessels’ biological structure and can be accepted into the body over time, empowering natural regrowth for a sustainable long-term solution.
Copying complexity
Blood vessels are strong but flexible. They are resilient and adaptable to contortion and changing conditions. These characteristics are granted by their structure and the materials of which they are composed. Concentric rings of elastin, a highly elastic connective tissue, alternate with layers of smooth muscle cells. Previous synthetic vessels have failed to mimic this arrangement, with permanent structures that don’t encourage natural regrowth or integration and tend to clog over time. Natural materials and specialised structure make a new class of artificial vessels a better prospect:
“These synthetic vessels are elegant because they are manufactured from just two naturally occurring materials that are well-tolerated by the body,” said Ziyu Wang, lead author and part of the research team from the University of Sydney. “Tropoelastin (the natural building block for elastin) is packaged in an elastic sheath which dissipates gradually and promotes the formation of highly organised, natural mimics of functioning blood vessels.”
The vessel provides a template for growth, and then fades away when the system has repaired, like removing the training wheels from a child’s bicycle.
Living artificial blood vessel
Passing control of the new arteries back to the body is the best way to ensure the treatment’s longevity but this has been impossible with previous synthetic vessels. In the current study, the researchers examined this process in mice, implanting the structure in the mouse abdominal aorta — the main artery carrying blood from the heart. Within 8 weeks, distinct natural cell profiles and surrounding structures had colonized the structure. After 8 months, mature fibres had taken hold and the artery had comparable thickness and performance to a natural, healthy aorta.
“Nature converts this manufactured tube over time to one that looks, behaves and functions like a real blood vessel,” said the study’s senior author, Anthony Weiss. “The technology’s ability to recreate the complex structure of biological tissue shows it has the potential to not only manufacture blood vessels to assist in surgery, but also sets the scene for the future creation of other synthetic tissues such as heart valves.”
Potential for growth
The integration of the artificial blood vessel into the natural system means it can grow and adapt with the body rather than present a rigid presence that becomes increasingly ill-suited over time. This is important for all patients, but is particularly significant for children, said co-author Christopher Breuer from the Center for Regenerative Medicine at Nationwide Children’s Hospital.
“Currently when kids suffer from an abnormal vessel, surgeons have no choice but to use synthetic vessels that function well for a short time but inevitably children need additional surgeries as they grow. This new technology provides the exciting foundation for the manufactured blood vessels to continue to grow and develop over time.”
This versatility to any future changes in the body makes these manufactured vessels an ideal solution to damaged vessels. The benefits of these flexible, adaptable vessels with perfected elastin structure could stretch beyond immediate improved surgery outcomes, to long-term improvements to countless lives.
Reference: Ziyu Wang, et al., Rapid Regeneration of a Neoartery with Elastic Lamellae, Advanced Materials (2022). DOI: 10.1002/adma.202205614
Feature image designed by Ziyu Wang and illustrated by Ella Maru Studio