The oviduct (or fallopian tube) serves as an environment for gamete transport, fertilization and preimplantation embryo development in mammals. Although there has been increasing evidence linking infertility with disrupted oviduct function, the specific roles that the oviduct plays in both normal and impaired reproductive processes remain unclear. The mouse is an important mammalian model to study human reproduction. However, most of the current analyses of the mouse oviduct rely on static histology or 2D visualization, and are unable to provide dynamic and volumetric characterization of this organ.
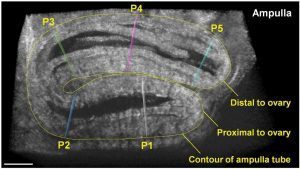
A frame from a 3D time‐lapse showing the ampulla tube and five positions (P1–P5) selected for analysis of the contraction wave
A team of researchers from the Baylor College of Medicine decided to address this limitation by demonstrating a 3D imaging approach that enables prolonged functional assessment of the mouse oviduct in vivo. By combining optical coherence tomography with a dorsal imaging window, this method allows for extended volumetric visualization of the oviduct dynamics, which was previously not achievable.
The approach is used for quantitative analysis of oviduct contraction, spatiotemporal characterization of cilia beat frequency and longitudinal imaging. This new approach is a useful in vivo imaging platform for a variety of live studies in mammalian reproduction.
The team presents a in vivo functional OCT assessment of the mouse oviduct through a dorsal imaging window, which allows for prolonged quantitative characterization of smooth muscle contraction and ciliary dynamics, as well as longitudinal analyses.
The 3D printed imaging window used in this study is inexpensive, easy to implant and convenient for prolonged and longitudinal imaging. Quantitative structural dynamics of the reproductive system, such as the velocity of the oviduct contraction wave, can be provided with this method as well.
“The major advantages of the presented method over the widely used bright‐field and fluorescence microscopy are the volumetric depth‐resolving capability and the non‐destructive mapping of CBF through the oviduct wall. This in vivo approach could be useful in studying dynamic aspects of normal reproductive events as well as investigating the effect of genetic and pharmacological manipulations” according to team member Irina Larina.