Molecular imaging provides a detailed glimpse into intricate biological processes and has emerged as a crucial tool in diagnosing numerous diseases and monitoring a patient’s response to therapy.
The development of optical contrast agents that are highly sensitive and specific to a particular disease, such as cancer, remains a great challenge for researchers. Small molecule optical contrast agents have demonstrated good biocompatibility for cancer imaging. For instance, indocyanine green (ICG), a cyanine dye, has been approved by the US Food and Drug Administration (FDA). However, poor solubility in water, aggregation-caused quenching, and rapid clearance from the body limited their application in clinic.
To overcome this disadvantage, one approach involves using polymers to improve the water solubility and extend blood circulation time of optical contrast agents.
A recent study published in WIREs Nanomedicine and Nanobiotechnology by Professor Xiqun Jiang of Nanjing University and coworkers, analyzes recent developments and challenges in the field of polymer-modified optical contrast agents to improve the sensitivity and specificity of cancer diagnosis.
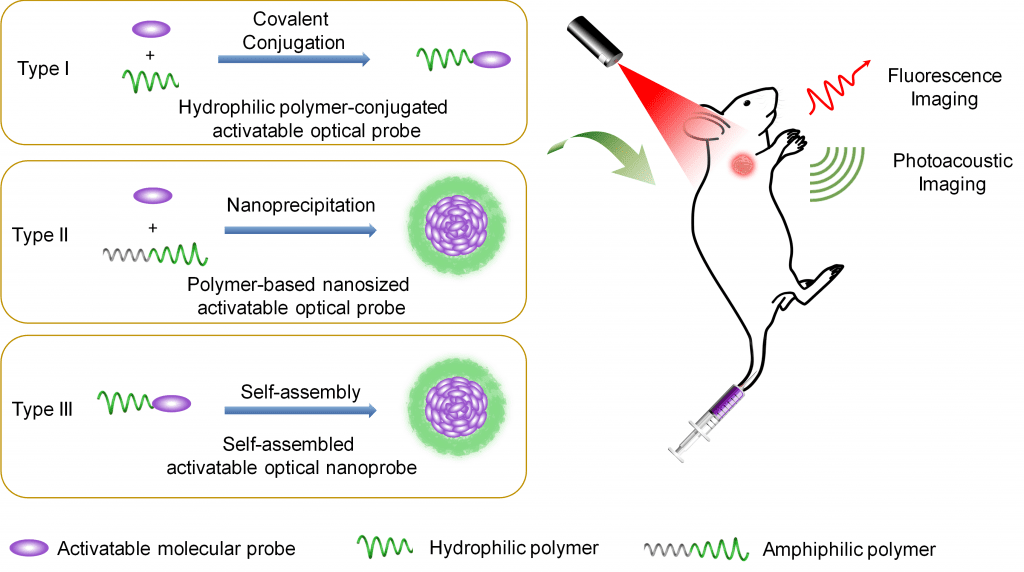
A number of hydrophilic or amphiphilic polymers linked to therapeutic agents have already been approved by the FDA for clinical use. For instance, polyethylene glycol (PEG), a polyether compound made from the condensation of ethylene glycol, is “generally recognized as safe” and can be linked through covalent or non-covalent conjugation of PEG polymer chains to small molecules or macrostructures, such as a drug or therapeutic protein. An successful exmaple of this is DOXIL®, the first FDA-approved nano-drug for cancer chemotherapy, and is comprised of PEGylated liposomal Doxorubicin.
Therefore, modification strategies which implement polymers to improve biological capabilities of therapeutics are also, logically, a promising avenue to improve the sensitivity and specificity of optical contrast agents. Polymer modification strategies create a hydrophilic layer around the optical contrast agents, endowing them with improved water solubility, and decreases the likelihood of proteins from binding to them in vivo, leading to extended blood circulation time.
Compared to “always-on” optical probes, which emit signals through accumulation regardless of their interaction with the target, “activatable” optical probes are designed in the “off” state and are designed to emit specific signals only in response to biomolecular recognition.
In such cases, the unique nature of the tumor microenvironment (TME) provides this opportunity as it differs from normal tissues and exhibits abnormal physiologic conditions, including weak acidity microenvironment, hypoxia, increased level of reactive oxygen and nitrogen species, as well as overexpressed enzymes. These conditions offer the potential biomolecular recognition sites to actuate the activatable optical probes with high signal-to-background ratio for cancer diagnosis.
While this field of research provides a promising strategy for developing enhanced diagnostic tools, challenges still remain before polymer-based activatable optical probes can be extended to clinical application. For instance, most polymer-based optical probes are cleared from the body via hepatobiliary excretion due to their relatively large sizes, leading to a longer retention time of probes compared to their small molecule counterparts.
Though the long-term safety of these polymer-based optical probes requires further investigation, this approach continues to offer the promise of significantly enhancing the sensitivity and specificity of cancer diagnosis to improve patient prognosis and, most importantly, survival rates.
Kindly contributed by the authors.