Scientists have proposed a new therapy that could potentially both detect and treat early stage Alzheimer’s disease, a complex neurological condition that affects over 50 million people worldwide.
“Early detection facilitates timely interventions, personalized treatment plans, and access to support services,” said Sudipta Senapati, a postdoctoral fellow in the lab of Shai Rahimipour at Bar-Ilan University, and first author of the current study that appeared in Small. “It also enables enrollment in clinical trials for testing more efficient disease-modifying therapies.”
Amyloid beta could be key to early diagnosis
The biology of Alzheimer’s disease, the most common cause of dementia, is generally characterized by the accumulation of two types of abnormal protein deposits in the brain: amyloid beta plaques that form outside neurons and tau protein tangles that grow inside them. Both routes disrupt normal cell function, leading to memory loss and neurological issues including epilepsy and the decline of cognitive function.
“In advanced stages of Alzheimer’s disease, the aggregation of beta-amyloid […] intensifies, resulting in the formation of amyloid plaques throughout the brain,” explained Senapati.
Most new therapies focus on targeting beta-amyloid aggregations to clear them away but haven’t shown significant improvements in human trials. One such example is aducanumab, a therapeutic monoclonal antibody that interacts with beta-amyloid aggregates and helps disassemble them.
Initially, experts were hesitant to approve it due to questionable effectiveness in clinical trials — while the reduction of the level of amyloid plaques in the brain was evident, the improvement in cognitive tasks wasn’t. About three years after its approval, the manufacturer decided to halt production.
According to Rahimipour, although these amyloid plaques are evident in late-stage Alzheimer’s disease, soluble forms of the protein, called oligomeric amyloid-beta species, pose a greater threat to nerve cells and cognitive function and appear earlier than amyloid-beta plaques.
For this reason, the team tried to find a way to detect soluble oligomeric amyloid-beta species in the brain. But this was not an easy task since during early stages of disease, they are present in low concentrations, which are difficult to detect.
Worms and sticky proteins
To fish for proteins, the team chose to work with a small, circular peptide called CP-2, which was previously known to interact with different oligomeric amyloid-beta peptides. Senapati and the team hypothesized that CP-2 would be able to “stick” to amyloid-beta proteins and prevent their aggregation that ultimately lead to the formation of the oligomers and plaques.
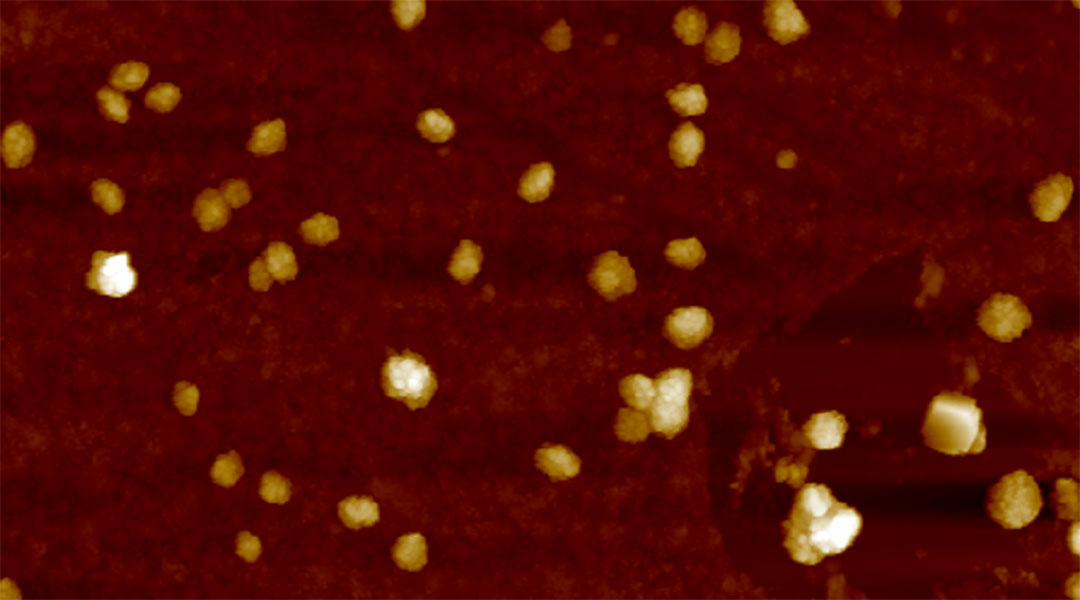
To deliver the CP-2 peptide into the brain, the team encapsulated them in small biological sacks called vesicles or liposomes that are made up of lipids and designed to stabilize and transport molecules, such as those used in some COVID-19 vaccines to carry mRNA. They also attached fluorescent proteins to helps track their movement in the body.
The team tested the viability of their therapy in worms called C. elegans, a conventional animal model for studying Alzheimer’s disease and other neurological diseases due to their simple nervous system, short lifespans, and genetic similarities to humans.
They found that treating sick worms with their vesicles restored a healthy lifespan (from 12.8 days back to a normal 14.6 days), and restored affected behaviors related to amyloid-beta accumulation, like the number of spontaneous movements the worms make per minute, which is decreased in the Alzheimer’s model.
But the next step was perhaps the most crucial. To be functional in humans — the point of developing such a therapy — the vesicles need to reach the brain. This, however, is not an easy task as it is protected by a biological barrier called the blood-brain barrier.
Getting into the brain
It has been previously established that the CP-2 peptide is itself able to cross the brain-blood barrier through an unknown mechanism, but Senapati had the added element of getting its vesicle carrier system, which helps keep the peptide stable, through.
Using a fluorescent imaging technique, that identifies fluorescence throughout the body, they observed fluorescence coming from the vesicles in the brains of treated mice. “While liposomes themselves have limited brain-blood barrier permeability, the conjugation of CP-2 on their surface significantly enhances this property,” said Senapati. She also explained that the team is currently investigating the exact mechanism by which CP-2 penetrates the barrier.
The team identified that fluorescence accumulation in the brain after four hours of treatment was significantly higher in diseased mice than in normal ones. Interestingly, in normal brains, the fluorescence went down as time passed, but in the brains of diseased animals, it went up after 24 hours and stayed steady for 72 hours following injection.
The team is investigating, but they suspect it is because the vesicles bind the amyloid species in the brain, taking longer to clear them, while in normal animals, they have nowhere to attach so they just clear out of the organ.
What are next steps?
This is significant as the result provides evidence that this approach could be used to detect developing Alzheimer’s in people who did have not yet shown any visible symptoms.
“Testing individuals for Alzheimer’s disease before symptoms is particularly crucial for those with specific risk factors such as advanced age (typically 65 years and older), family history of Alzheimer’s, genetic predisposition, cognitive concerns, or related medical conditions,” said Senapati.
Before this technology will make its way to clinical trials, Senapati explained the team needs to work on optimizing the vesicles. “The stability of liposomes under our experimental settings extends up to a month, providing a reasonable timeframe for drug delivery from the manufacturer to the clinic and ultimately to the patient,” she said. “However, the stability may be further optimized by adjusting the pH, medium, etc., requiring additional experimentation and refinement. It should be noted that liposomal drugs are widely available for treatment of many diseases and symptoms with expectable stability.”
Moreover, as the study showed vesicle accumulation in liver, heart, spleen, kidney, and lungs, “In next-generation [vesicles], liposome surface will be modified with known approaches to reduce accumulation in these organs,” he added.
“This innovative strategy holds potential as a breakthrough in the battle against Alzheimer’s disease, offering a ray of hope for early detection and targeted therapy,” concluded Senapati.
Reference: Sudipta Senapati, et. al., Multifunctional Liposomes Targeting Amyloid-β Oligomers for Early Diagnosis and Therapy of Alzheimer’s Disease, Small (2024). DOI: smll.202311670
Feature image: Atomic force microscopic image of the liposomes