Humans have always been curious whether they are indeed alone in the Universe. While the jury is still out on this existential cosmic question, it turns out that we are never really alone because the human body is technically a universe of its own.
More or less unbeknownst to us, our body is able to host a bustling community of microorganisms, such as bacteria and fungi, for whom we are their whole world. They form different microbiomes, tiny, cooperative ecosystems found in locations such as the skin, the mouth, or the gut.
Many of these microorganisms work for the benefit of the human they inhabit, helping us keep pathogens in check or digesting the food we consume. However, peace is not an option for some, and it is important to investigate how the good, the bad, and the ugly interact with each other to avoid the onset of serious diseases.
Investigating microbiomes is tricky because recreating its specific niche and successfully populating it with the resident microorganisms is incredibly difficult. Organs-on-a-chip are tiny devices populated with different cells in order to mimic the physiology and environment of any given organ. The simplicity of the organ-on-a-chip system creates the perfect environment to investigate the different interactions between microorganisms that reside within the human gut.
For this purpose, engineers Chwee Teck Lim and Jee Yeon Lee from the National University of Singapore created an organ-on-a-chip that works as an artificial gut, which they’ve used to gain deeper insights into the biome that exists there, and how dysregulation can lead to disease.
Lim and Lee sat down with us and explained the fascinating world of microbiomes and the important results their newly conceived gut-on-a-chip has produced.
Can you tell us what microbiomes are and where they can be found on or in the human body?
Chwee Teck (CTL): The microbiome is kind of like a collection of microorganisms. It can include not just bacteria, but also fungi, viruses, and others present throughout the body. In the context of the human microbiome, this refers to a diverse array of these microorganisms distributed in various regions, like our digestive track, respiratory system, skin, mouth, and even the genital system.
What is the function of these microbiomes and how much do they vary between individual human beings?
Jee Yeon (JYL): The role of these microbiomes is not entirely clear yet. Over the years, many researchers have found that they are connected to human health and disease. For instance, gut microbiome helps us to digest food and regulate our immune system and even produce vitamins, like B12. Scientific articles have found the microbiome can be linked to many diseases such as cancer, inflammatory bowel disease, metabolic disease, and even neurological disorders. Despite these connections, there’s still much we don’t understand about what exactly they do.
What’s interesting is that human microbiomes differ from person to person. Your gut microbiome can be pretty unique because it gets influenced by many things- your diets, age, the environment you’re exposed to, your overall health, genetics, the medications or antibiotics you use, and even where you live. You could way it’s our very personal and unique microbial fingerprint.
Can microbiomes change depending on human behavior?
CTL: This is a very interesting and fascinating question which motivated us to want to study this topic. We do play a significant role in shaping our own health based on our behavior. What we eat, for example, affects the dynamics of the gut microbiome significantly. Our choice of food and also our medication plays a part in shaping the microbiome in our guts. If you look at some of the studies, factors such as exercise, stress, and mood also have some influence and causes this constant fluctuation of the microbiome in our gut.
With the diet example, the choice of whether we take in vegetables or meat impacts the microbiome. Some parts of the microbiome feed on fiber, but if you significantly reduce the intake of fiber, that set of the microbiome tends to decrease. When we take antibiotics, that also changes our microbiome composition significantly because a lot of the bacteria are killed by the antibiotics. So yes, the microbiome itself is highly responsive and undergoes continual changes based on our behavior throughout our lives.
There is this interesting example of women giving birth naturally versus giving birth via a C-section. During natural birth, the baby is literally being coated with vagina bacteria, which helps in contributing towards the health of the infant much more than a C-section.
In your recent study, you investigated the interaction of different bacteria of the human gut. What is special about the gut microbiome and why is the investigation important?
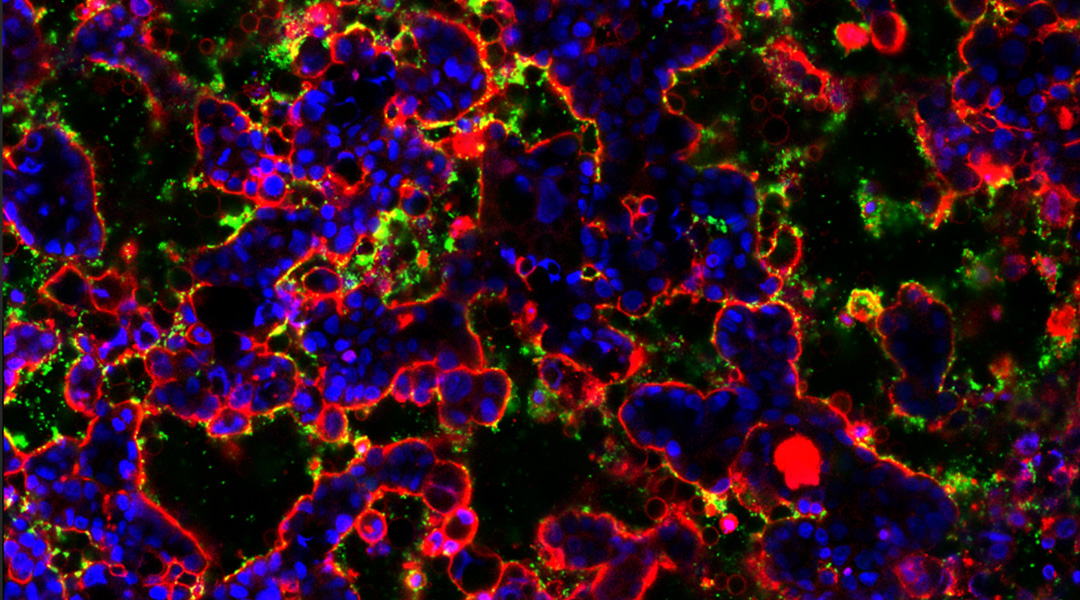
JYL – In this study, we investigated two types of gut bacteria with distinct functions. First, enterotoxigenic Bacteroides fragilis (ETBF) gut bacteria, one of the three bacteria known to be carcinogenic and linked to colorectal cancer. The other bacterium we used is the common probiotic, Lactobacillus, which plays a role in strengthening the intestinal barrier. We aimed to explore the interaction between these two bacteria and their influence on gut health.
When we initially used the enterotoxigenic Bacteroides fragilis (ETBF) alone, it disrupted the healthy state of our gut model. ETBF produces genotoxin [an agent that causes DNA damage] and exhibits pathogenic behavior, disrupting the 3D structure and the barrier function in the gut. We also observed the bacteria initiate several tumorigenic signaling pathways.
When we pretreated the gut model with probiotic bacteria before adding ETBF, similar to our gut covered with commensal bacteria existing since birth, they acted as a protectors against the pathogenic bacterium through competition. We observed that our gut model treated with this beneficial bacterium remained very healthy and was not affected by the pathogenic behavior of the harmful bacteria.
What excites you most about this work?
CTL: We have successfully developed [a chip] that can recapitulate the essential features of environmental gut in a scalable and reproducible manner and also enabled the culture of the diverse gut microbes that inhabit our gut.
Achieving that took about four or five years but I am glad that we did it eventually. I think second is that our chip also enabled us to observe the behavior of gut microbes, resulting in a [realistic] 3D microenvironment. I think that is important because you want to understand how these microbes interact and from that deduce how they give rise to diseases or offer protection to the gut in the present state of a pathogen.
What kind of device is your chip?
JYL: The gut microbiome chip serve as a co-culture device, facilitating the co-culture of the 3D biomimetic gut with all types of gut microorganism together. A key feature that sets our chip apart is its ability to visualize gut microorganisms within the 3D gut using high-magnification imaging, enabling identification of various microbial species, and the monitoring of their behaviors, as well as the gut’s response through real-time imaging or assay.
What materials do you use to build this device?
JYL: Our device uses a very standard method called photolithography and soft lithography to generate microfluidic device. Photolithography is a technique that uses UV light to create molds with micrometer precision. Soft lithography is a technique that uses soft materials like silicones or other polymer material to replicate the patterns from these molds for rapid prototyping.
We first designed and created the pattern to make a master template using photolithography on silicon wafer. On that master template, we pour a transparent and biocompatible elastic polymer, which is PDMS silicon polymer, onto the mold and peeled out the replica with the negative design of our master mold. This process is similar is similar to creating a mold for cookies, baking them with cookie dough using a mold. The procedure is very straightforward and efficient. Due to our highly scalable design, we can easily fabricate 100 chips per day.
How long can you keep the health of the cells and is the lifespan comparable so that you can really mimic the human microbiome for your experiments.
JYL: In our system, we undertake two key steps : initially establishing the gut model and subsequently introducing gut bacteria to form a co-culture. In the first step, we build a 3D biomimetic gut, starting with the culture of human Caco-2 cells. These cells initially form a monolayer and subsequently develop into a 3D structure under mechanical stress. The process typically takes about five to six days until the lining is fully developed. In the second step, we introduce bacteria to observe their behavior within this 3D gut system.
We were successful in culturing the gut model without bacteria for up to 10 days. However, we limited the incubate time intentionally, as our primary goal was to establish a rapid gut model, minimizing duration required for studying the gut microbiome. This approach contrasts with other conventional systems, like organoids, which typically take several weeks to nearly a month for proper assembly and result delivery. Efficiently investigating the gut microbiome is generally challenging, but we were able to complete our experiments in comparatively short amount of time. With continued regular nutrient supply, we might extend the lifespan of the cells in our system for a few weeks.
Our gut model closely mimics the actual human gut in several key aspects including the formation of a 3D structure, the presence of differentiated cell types, the exhibition of mucin secretion and protease activity-resembling the structure and functions of the human intestine. However, there are inherent limitations to cell-based gut models in fully recapitulating the intricate complexity of the human intestine, representing common challenges in organ-on-a chip system.
Nonetheless, our system provides a highly efficient and advanced platform compared to current methods in establishing gut-microbe interface and studying their behaviors. This technology can help us to investigate microbe-induced gut diseases and develop novel therapeutics.
Your organ on a chip can also be used for experiments for live cell imaging. How do you apply your device in this context?
CTL: This is the key advantage of our chip; having the ability to visualize the gut micro interface in the living state. When we were doing our literature search, we found that there were other guts on chip devices that were limited in what they could image in a 3D environment.
We wanted to develop a device that enables us to do dynamic monitoring of both living bacteria and living gut cells. This also includes observing bacterial growth and then identifying the beneficial behavior from the bacteria and trying to capture the interactions among the different species of bacteria. Our chip can also enable monitoring of the cellular response of the gut to this microbial interaction, basically observing the actual interaction with real time monitoring between the microbe species and the micro gut cells.
Is there a specific method you use?
JYL: Our chip offers a crucial advantage by allowing the visualization of the gut-microbe interface in a live state. This encompasses the observation of bacterial growth, identification of pathogenic or beneficial behaviors, and capturing interactions with other species. Furthermore, our chip facilitates the monitoring of dynamic cellular responses in the gut to these microbial interactions. In our experiments, we employ imaging techniques and analysis to identify and observe the behavior of gut bacteria as they colonize the gut. For imaging, we commonly utilize confocal microscopes and real-time microscopes to visualize bacteria, given their small size ranging from 1.5 to 2 microns. However, the device can seamlessly integrate with any type of lab microscopy.
What are your future plans for your gut microbiome on a chip?
CTL: Our future plans involve advancing both the engineering and the biological aspects of the device. From the engineering perspective, we aim to see how we can further refine the device by mimicking the complex microenvironment in the human intestine. For example, we might incorporate more complex mechanical cues and enable or enhance the cellular complexity using gut organoids. Right now, we use Caco-2 cells, which is a cell line that originated from a colon carcinoma. Now, maybe we could use gut organoids, which are more complex but also more representative of what is inside the gut itself.
In terms of biology, our goal is to introduce a complex microbial community to better understand how they assemble, interact, where do they localize within the gut, and how they behave under various stimuli like diet or antibiotics. We originally created the gut-on-a-chip to understand how nutrients get absorbed into the gut.
Right now, in current studies, someone eats food and then you analyze the stool, but we do not know what is happening inside the gut. Hopefully our gut chip can help us better understand how certain nutrients get absorbed and whether those microbes may interfere with the absorption of nutrients. We are trying to understand how this microbial community will influence our gut health essentially. We want to advance our understanding of the gut microbes’ role in our health and disease.
Do you envision in the future that you have personalized gut microbiomes on the chip and testing how individual got microbiomes are responding to different treatments?
CTL: We are looking at either preventive medicine or personalized medicine because each of us have different sets of microbes. Some scientific publications right now talk about how microbes may interfere with or assist in chemotherapy treatment. Two patients who get the same treatment end up with very different outcomes. This research boiled down the difference to the actual microbes in the gut because some microbes may be targeted to improve efficacy and reduce the toxicity of chemotherapy treatment.
We all have different microbiomes, so maybe we can personalize our own gut avatar and run experiment to see what drugs will work and what drugs will not. There are implications for the development of microbiome-based therapeutics.
Reference: Chwee Teck Lim, et al., Dissecting Gut-Microbial Community Interactions using a Gut Microbiome-on-a-Chip, Advanced Science (2023). DOI: 10.1002/advs.202302113