With the recently increasing development of lightweight, portable, flexible, and wearable electronics for diverse health and biomedical devices, there is an urgent need to explore new power sources that offer higher flexibility and human/tissue-adaptability for these electronic devices. Researchers have engineered a next-generation battery technology, known as metal–air batteries, which can be easily fabricated into flexible and wristband-like cells. Although devices powered by metal–air batteries still have a way to go before they are ready for commercialization, the current studies have established solid evidence that these devices can provide enormous opportunities for the development of the next generation of flexible, wearable, and bioadaptable power sources.
“Theoretically, neutral-electrolyte-based Mg–air batteries possess potential advantages in biomedical applications over other alkaline-based metal–air counterparts,” says Dr. Chong Cheng, an AvH research fellow and carbon nanomaterials specialist working in the Department of Chemistry at Freie Universität Berlin (Germany). However, the conventional application of Mg–air batteries has faced several challenges to be overcome, one of the most important scientific difficulties that reduces the performance is the sluggish kinetics of the oxygen reduction reaction (ORR) in the air cathode. Currently, the rational design of advanced oxygen-electrodes for Mg–air batteries with high discharge voltage and capacity under neutral conditions remains a major challenge within the research community. The scalable synthesis of carbon-based oxygen electrocatalysts integrated with high ORR catalytic activity, open-mesoporous and interconnected structures, and 3D porous channels for the air cathode has yet not been realized.
A new strategy to overcome the current limitation on sluggish reaction kinetics of the air cathode in Mg–air batteries from Dr. Chong Cheng and Prof. Rainer Haag at Freie Universität in Berlin describes the scalable synthesis of atomic Fe–Nx coupled to open mesoporous N-doped-carbon nanofibers as an advanced oxygen electrode for Mg–air batteries. Inspired by the fibrous string structures of toadspawn, they designed a novel fabrication strategy based on the electrospinning of polyacrylonitrile-branched silica nanoaggregates solution and a secondary coating and carbonization of a thin layer of an Fe-doped zeolitic imidazolate framework; “these endow the fabricated carbon nanofibers with an open mesoporous structure and homogeneously coupled atomic Fe–Nx catalytic sites” said the researchers.
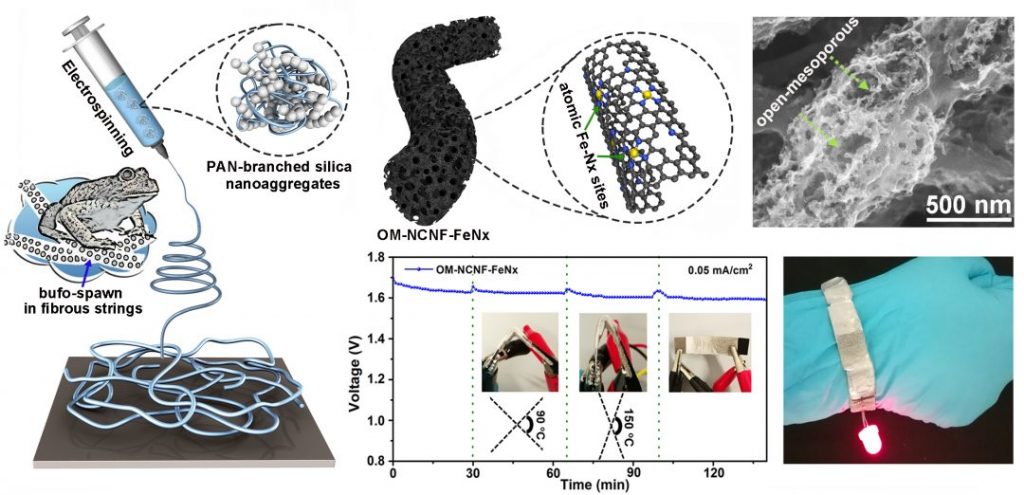
Design of an air cathode for metal–air batteries, inspired by toadspawn
The obtained oxygen electrocatalyst and the accordingly constructed air cathode show manifold advantages, which include interconnected structures and 3D hierarchically porous networks for ions/air diffusion, good bioadaptability, and high oxygen electrocatalytic performances for both alkaline and neutral electrolytes. Most importantly, the assembled Mg–air batteries with neutral electrolytes reveal high open-circuit voltage, stable discharge voltage plateaus, high capacity, long operating life, and good flexibility.
There’s still some time before Mg–air batteries will help to power commercial electronic and biomedical devices, but now that future appears a bit closer. “We believe that this novel and advanced oxygen electrode can meet the challenges and urgent needs for efficient air cathodes in Mg–air batteries with neutral electrolytes, but more work is still needed,” said Prof. Haag. Furthermore, the authors believe that this study not only creates many opportunities in the future design of metal–air batteries to achieve flexible, wearable, and bioadaptable power sources for diverse electronic and biomedical devices, but may also open new pathways for the scale-up production of advanced nanofibrous carbon electrodes for a broad range of applications wherein fast ion/electron storage/transportation, metal-atom doping, and cytocompatible electronic-cellular-interfaces are highly required, such as for Li-ion batteries, supercapacitors, electronics, biosensors, and even tissue monitoring and regeneration.












