The quote, “Every tooth in a man’s head is more valuable than a diamond”, must ring very true for biochemist Robert Layfield and Anisur Rahman, an expert in autoimmune rheumatic diseases and chronic pain.
It was the teeth found in old skeletons and a curiosity for what they might contain that brought these scientists from two very different fields together for a fascinating endeavor.
Every tooth consists of various layers — within the tooth lies the dental pulp, which is made up of blood vessels, nerves, and different tissues, while the outside of the tooth is covered by the protective enamel. The interior of the tooth can be preserved for extended periods of time and provide a plethora of archeological information, such as the individual’s age, sex, ethnicity, and, potentially, the diseases they suffered from.
Previous studies have shown that the teeth of deceased individuals can still harbor intact red blood cells, DNA, RNA, and various other proteins. In fact, scientists were awarded the Nobel Prize for Physiology and Medicine last year for their work in extracting ancient DNA from bones, which has greatly contributed to our understanding of our past.
However, what captivated Rahman and Layfield was the possibility of finding intact antibodies, complex proteins generated by the immune system to bind and neutralize pathogens as their presence would offer an opportunity to probe for a distinct past infection as a result of their “molecular memory”.
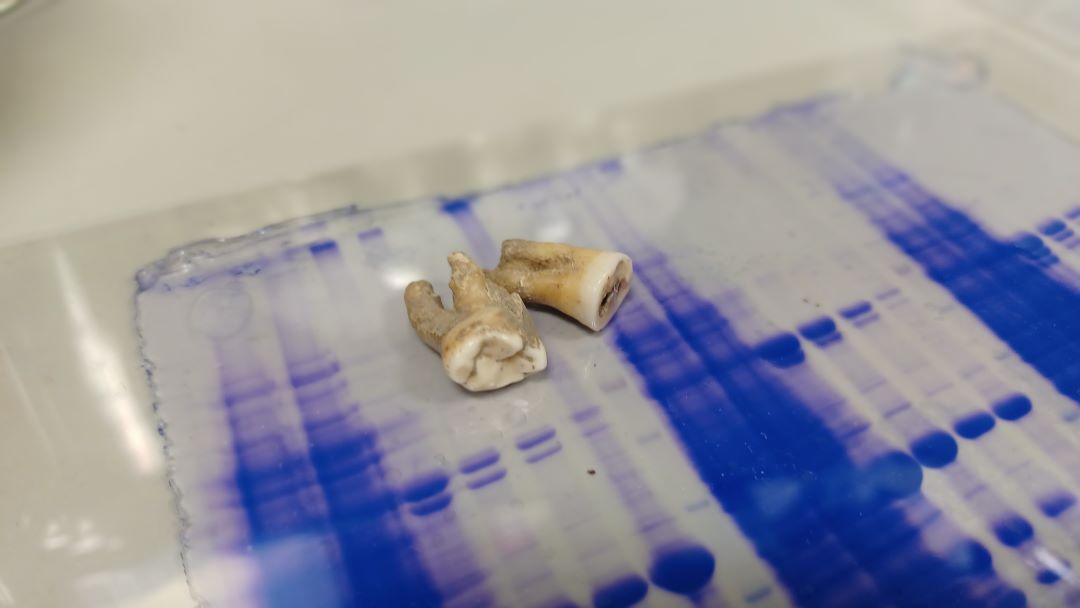
The duo has recently published an intriguing paper in which they isolated fully functional antibodies that were frozen in time for roughly 800 years and yet are still able to bind a virus that infects many humans today.
Rahman and Layfield took the time to bring us along on this fascinating journey, exploring the history of humanity and the pathogens that have plagued us for centuries.
In your recent research paper, you work with medieval human teeth extracted from skeletons. Where did these skeletons come from?
Robert Layfield (RL): They are from the Chester Greyfriars Collection from the Northwest of England. The samples were available through collaborators at Liverpool John Moores University and these teeth date back between 1285 and 1470 CE.
Anisur Rahman (AR): The real story here is two people and two groups who would never have met each other and would never have worked with each other from completely different fields somehow coming together coincidentally.
Rob and his group had been interested in ancient bone diseases, and they published a paper on a medieval disease, which is a disease of the bone where you would see abnormalities in the skeleton of those affected, so you could reliably prove that a skeleton had Paget’s disease.
RL: Subsequently my group studied a different skeleton with some etchings on the facial bones that showed that the bone had eroded away. We asked ourselves which diseases could potentially affect the face, and one possibility was systemic lupus erythematosus, also known as lupus.
Layfield’s team therefore contacted LUPUS UK, a patient charity in the United Kingdom, asking if they knew anybody who might be able to help them, and so LUPUS UK got him in touch with Rahman’s group.
AR: It would have been easy to say no because we were fairly confident this was not lupus, but my colleague Thomas McDonnell and I thought it was very interesting and would not do any harm to find out more.
We did not think it was likely to be lupus because the pathology of that disease is that the rash is very superficial. It could never eat through the face into the bones. However, we could, if necessary, test the binding properties of antibodies extracted from the skeleton. That brought us to ask if we could prove antibodies in bone samples have the ability to bind antigens at all.
Are there ethical restrictions you have to take into consideration? Was it explicitly defined from which part of the body you could take samples?
RL: The study of these skeletons is not covered by the ethical restrictions in the UK that involve the Human Tissue Act, which controls tissues from present day patients. Nevertheless there are guidelines and very clear guidance as to handling of human remains, although they are hundreds of years old.
The reason we work with teeth is that archaeologists seem to be happier to share teeth. It is less destructive to the overall remains to pull the tooth out and also, they can be replaced with models. It is really about convenience, and it was not necessarily planned that the tooth is the place to go looking for these things.
We really just need any skeletal tissue with a good blood supply because we are presuming these antibodies come from blood that has been captured. In another study with our mammoth samples, we were able to get antibodies from femur bone as well. We would reason that any mineralized tissue with a good blood supply is a likely target to find antibodies.
AR: I would say that the mouth is a very immunologically active area where you would expect many antibodies to be present, so if you gave me a choice, I would prefer teeth over bone.
Are there specific teeth that work best?
RL: The important thing is blood supply, so a molar might have a slightly better blood supply than other teeth, but you must test with different teeth to know.
You suspected that these skeletons had diseases. What were you looking for and why?
RL: It is not unusual in the archaeological record to find signs of disease, like osteoporosis or arthritis. We were selecting conditions where we might anticipate this sort of alteration in immune signaling or in the immune system as well. Two of the skeletons we looked at had evidence of rheumatoid arthritis and osteoporosis which, again, is pretty common in the archaeological record and you might anticipate immune dysfunction.
AR: It is not so much immunity because osteoporosis and Paget’s disease are not really immune diseases. They are diseases which leave their mark on bone. You are not going to find a mark on bone with chickenpox or malaria, for example.
About ten years ago, I was involved in a similar project where they dug up a skeleton and they were convinced it had rheumatoid arthritis. The interesting thing was rheumatoid arthritis was not supposed to exist in Europe prior to the 1300s, and this skeleton was older. They thought they found the earliest rheumatoid skeleton and so they sent us all the X-rays and samples and we got a pathologist and a radiologist to examine the data and it was not conclusive. We would have loved to say these were characteristic erosions of rheumatoid, but it was not clear enough. Lupus, which I specialize in, can have effects on bone, but it’s much rarer.
RL: Anisur mentioned at the start about our research on ancient Paget’s disease, which is a bone disorder. We found some of the disease-associated protein changes in the teeth of medieval skeletons with this disease and that discovery moved us from bones to teeth.
The reason we considered teeth was because, in parallel, there was an ancient DNA analysis performed on teeth samples and there was leftover material from the teeth. That was really quite unexpected because Paget’s is a bone disorder and teeth are not bone, yet there are pathological protein changes in teeth that mirror those seen in ancient bone afflicted with Paget’s disease.
How did you sample the teeth?
RL: We start by grinding the teeth with just a pestle and mortar, and there are standard protocols for ancient protein extraction. Then the protocol involves decalcification, removing the calcium or calcium phosphate, and that just leaves protein. It is a very simple and very crude sequential extraction, albeit under clean conditions. That purified fraction went to Anisur and Thomas at UCL as they have the expertise to actually extract functional antibodies from those crude protein extracts, which themselves contain hundreds of ancient proteins that are preserved.
AR: You need to focus on what you might be interested in because there are just so many proteins, and in our case, we focused upon antibodies. I suppose if you had enough of the sample, you could run it through a mass spectrometer see what you have there, but I am not sure that is part of the plan at the moment. We were looking for particular antibodies rather than looking at everything.
RL: Other groups have noted the presence of antibody sequences for quite a long time in these ancient protein extracts, but no one had really asked if there were whole antibodies present. There was actually a paper in the literature that suggested that antibodies were poorly preserved and that was part of the motivation for wanting to test that idea ourselves.
Could you describe what antibodies are in general terms and why you were looking for them in particular?
AR: Antibodies are the key molecules of our immune system, which protects us against diseases. Antibodies are very specialized molecules which can recognize foreign proteins like bacteria, viruses, parasites and so on. Antibodies are made up of four chains, two heavy and two light.
The clever thing about them is that these chains can vary in sequence such that each antibody has a particular job. You can have specific antibodies against the flu, Epstein-Barr, or COVID-19, for example. They are designed to recognize and attack foreign organisms.
The question was if there were just bits of the antibodies, like one chain or fragments of chains, or were there whole antibodies. In our group, we do a lot of purifying antibodies from patient samples on columns, which are very good at dragging antibodies out of a mixture, and that is what my colleague Thomas did. You can tell whether the antibodies are complete molecules or not by various different biochemical techniques like running them down gels, seeing what size they are, and reducing them chemically to see if they break into heavy and light chains. You will see in the paper that there are gels showing that the antibody from the ancient skeleton is located in the same place on the gel as a modern human antibody run next to it.
That was the proof antibodies exist in a whole form, but that does not prove they are still functional. To prove that they are functional, you have to prove they still bind something.
There was every chance this whole thing would never have been published and every chance that we would not find whole antibodies in these samples. At each stage, as the steps kept working, we simply moved on to the next step. It eventually came back to Rob’s team, and he wanted to test these antibodies against something and see if they bind.
RL: Anisur helped choose the Epstein–Barr virus as a pathogen we would expect most antibodies to react with and so we used a technique called western blotting, which is a pretty standard protein chemical technique for showing functionality of antibodies.
How does western blotting work?
RL: Western blotting is a very commonly used analytical technique which protein chemists apply to detect the presence of specific proteins in a biological sample. In that case, the antibody is used as a tool to not only confirm the presence, but also get information on size and modifications of the protein.
We turned things on their head and we used the western blot simply to show that the purified ancient antibodies still had reactivity with a positive signal on the western blot. That involved taking an Epstein–Barr virus protein, separating it on an electrophoresis system, and then trying to detect that protein with our antibody.
The positive signal that we saw told us the antibody still retained its ability to bind to one of its viral targets; one of the Epstein–Barr virus proteins.
How is it that these antibodies were still intact and well-functioning after all these years?
AR: I would turn that question on its head. Why should the antibodies not be functional? They should not be exposed to proteases or reductases [compounds which digest antibodies] because they are sitting inside the tooth. Essentially it is a locked room inside the tooth where nothing dangerous to the antibody can get inside to digest the antibody.
RL: Within that conversation, there is some evidence that calcium phosphate, which is the main inorganic component of teeth and bones, is itself protective towards proteins. The mechanism probably is that it helps exclude water and enzymes such as proteases that need water as a cofactor to break down other proteins.
The other thing to think about is proteins are built from amino acids and they are built with peptide bonds. The bonding in the molecules themselves is covalent bonding and these are really strong bonds, so a few thousand years on an evolutionary timescale of millions of years is not really long. Some of the work in the field has shown six- or seven-million-year-old proteins being preserved in other calcified materials.
AR: I think the calcification is very important and one of the many lovely things about this research is, although Rob says it is not surprising, it is completely counterintuitive. You would never have guessed it. I am thinking if five years ago, before I met Rob, if a medical student had asked me if antibodies can survive hundreds of years, I would say that is very unlikely.
RL: Incidentally, when I started my research career, probably 30 years ago, our consultant neuropathologist came to see me with brain tissue that had been preserved in formaldehyde for two or three years. He asked me to analyze proteins from this material.
As a chemist by trade, I said absolutely not, because that is a chemical that crosslinks proteins. I ended up trying anyway, and found amyloid proteins, which are disease-associated proteins, that were incredibly well preserved. I think this idea of always having an open mind is really one of the lessons reinforced by our study.
Why would antibodies against Epstein–Barr virus be present in the teeth?
AR: To be honest, in the Twitter feed people have ascribed all sorts of clever ideas to why we chose to test Epstein-Barr virus but in reality, it was just our best guess. I thought, we know absolutely nothing about these skeletons, and we do not know what infections they have been in contact with. If you give me a random sample from a human being today and ask me what antibodies I think the human being might have, I would go with Epstein–Barr virus because that has infected about 90% of human beings.
I did not know for sure whether that was still true in the 12th or 13th centuries, but I cannot think of another virus which has such a high prevalence of antibodies against it. Rob’s team then said they could buy the antigen for Epstein–Barr commercially and test for any reactivity. It was nothing more complex than our best guess.
RL: I would predict they are just a sample of the circulation and there will almost certainly be antibodies against other pathogens present in the samples.
What does the detection of contemporary Epstein–Barr virus antigens mean concerning virus evolution?
AR: I just think that Epstein–Barr is probably a conserved virus, and it was around in those times, although I do not actually know if Epstein–Barr was officially recorded as being around back then.
There is a whole science of historical medicine and people trying to work out whether particular figures from history had particular diseases. I once was sent a paper to review trying to prove that Jane Austen, the famous author, had lupus. I ordinarily would not have accepted this review invitation, but I really love Jane Austen and I was curious how are they were going to prove this.
What they had done was read her diaries and they picked up on her descriptions of waking up with a terrible rash or fatigue and they put this together into an amazing story that she must have had lupus. I rejected the paper because, although interesting, there was really no science in it at all.
Could any type of antibody present at the time of death be isolated from teeth centuries later or are there restrictions on what you can find?
RL: Theoretically, no. There are no restrictions other than presence in the first place. There are other studies that have used other immunological approaches to try and find evidence of ancient antibody reactivity, and they do point toward the conditions being critical for sample preservation. It would be conjecture, but I think it is entirely feasible that other disorders could be detected with antibodies.
Someone approached me recently to ask about English sweating sickness. This was a disorder, as I understand it, where you started sweating and 30 minutes later you were gone. Could that have been caused by a virus?
AR: There was some time in the 1530s when a lot of London had the sweating sickness. People would send their children out to the countryside and evacuate them from London, so it is a well-known disease but as far as I know nobody knows what the organism was that caused it.
Would it be possible to extract whole infectious agents from the teeth?
AR: Apart from viruses, infectious agents are living things so I do not think a bacterium would live for hundreds of years because it needs energy to survive and divide. Maybe you could have an intact virus particle, but I do not know enough about dentistry to know for sure.
You also explored the potential to isolate antibodies from a mammoth femur. Are you planning to look for evidence of other antibodies or even infectious disease in skeletons in the future?
RL: We would love to. I think that truly interdisciplinary research and getting funding to do that is still new and a bit of a challenge. The landscape is changing however, and the UK is launching specific schemes that are directed towards genuine interdisciplinary research.
AR: I was thinking about looking at the cause of the Athenian plague. It is about creating a good story and getting access to the materials. For example, I was thinking of talking to the department of Classics at UCL to see what sort of collections they have.
My son did a classics degree and he learned about the plague of Athens, which was in the 5th Century BC, and which was written about by Thucidydes, and nobody knew what caused it. If you could dig up some skeletons from those times, you might be able to find out which pathogens they had antibodies to and that would be very interesting.
RL: We are hoping publicity will attract the archaeologists, though certainly basic scientists and archeologists work in very different ways and archaeologists tend to be understandably protective of their collections, but we would love to be approached.
Feature image credit: Robert Layfield