 From car exhaust pipes to the pharmaceutical industry, catalysis plays a central role in most of the chemical processes that have a direct impact on our daily lives. The development of new, more efficient and inexpensive catalysts is directly linked to the effort to overcome current challenges such as environmentally harmless energy production.
From car exhaust pipes to the pharmaceutical industry, catalysis plays a central role in most of the chemical processes that have a direct impact on our daily lives. The development of new, more efficient and inexpensive catalysts is directly linked to the effort to overcome current challenges such as environmentally harmless energy production.
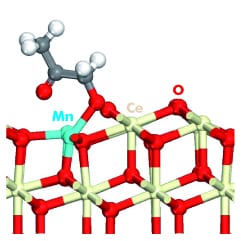
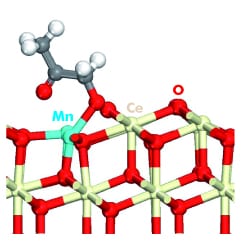
An important group of solid catalysts are metal oxides. Among them, ceria, CeO2, based catalysts have shown promise for relevant reactions such as CO or CH4 oxidation, or sulfur adsorptive desulfurization. Doping of ceria with the addition of other transition metals into its lattice, often substituting cerium atoms, can improve its catalytic properties. The potential of these catalysts is driving extensive experimental and theoretical research.
In metal-doped ceria catalyzed processes the surface cycles between oxidized, partially reduced and reduced conditions. Therefore, theoretical modeling of the catalytic activity of these materials needs to accurately reflect the energetics for transformation along these cycles.While Density Functional Theory (DFT) is widely employed in catalysis studies, its use with transition metal oxide catalysts in general and ceria-based catalysts in particular is limited by the inability of conventional DFT to correctly represent localized, strongly correlated electrons, such as the ones in the localized f-states which are fundamental to explaining the properties of ceria-based catalysts.
The most common method for correcting the error produced by conventional DFT in the representation of strongly correlated electrons consists of adding a semiempirical term, the Hubbard U, to the localized states (DFT+U), as described in detail in a review article by Himmetoglu et al., published in the Int. Journal of Quantum Chemistry this year. U is very frequently chosen to match known properties of the material.
However, this may not necessarily give correct surface reduction energetics. Another option is the self-consistent calculation of U, but it does not return the full ab initio nature to DFT when the self-consistent U-values change along the reaction path. In this case, a constant U needs to be empirically chosen to allow the comparison of the system energy along the catalytic cycle. As an alternative to the DFT+U method, a hybrid exchange-correlation functional can be used. It combines a specified, empirically determined amount of nonlocal exact exchange with a conventional DFT functional. This improves the capacity of DFT to deal with localized electrons but heavily increases its computational cost.
Now, new work by Krcha and Janik (Pennsylvania State University, US), also published in the Int. Journal of Quantum Chemistry, discusses the specific challenges of applying these methods to the modeling of the surface catalytic properties of transition metal-doped ceria. In the case of DFT+U, these challenges include the U-value determination for cerium, the role of the transition metal dopant atoms in the catalytic process and the necessity, in certain cases, of adding a U value to the d-states of the metal dopant and/or the p-states of oxygen. An additional complication comes from the fact that a different U potential may be needed for the same atom in different geometric arrangements. The use of a position dependent version of DFT+U (DFT+U(R)), already used with other materials, is presented as a promising approach.
Regarding hybrid exchange-correlation functionals and their high computational intensity, this method is suggested for smaller systems to help calibrate U values for DFT+U calculations. Standardization of current “best practices” to help compare different studies as well as a greater availability of experimental model systems for benchmarking are the proposed route to further development in the modeling of these important materials.