 Natural polymers are attractive agents for improved delivery, stabilization and prolonged release of encapsulated biological molecules. Amongst the range of biomaterials used to date, alginate-based hydrogels represent one of the most promising candidates owing to their biocompatible nature and fast biodegradability.
Natural polymers are attractive agents for improved delivery, stabilization and prolonged release of encapsulated biological molecules. Amongst the range of biomaterials used to date, alginate-based hydrogels represent one of the most promising candidates owing to their biocompatible nature and fast biodegradability.
It is not a surprise, therefore, that significant efforts have been dedicated to produce alginate hydrogel particles for various biomedical and pharmaceutical applications. However, production of small size particles approaching the dimensions of a cell has been challenging due to the relatively high viscosity of alginate solutions and poor control over the cross-linking kinetics. As a result, most of the microfluidic methods employed to date generated relatively large particles of 50-100 µm in size, thus limiting further applications in biomedicine.
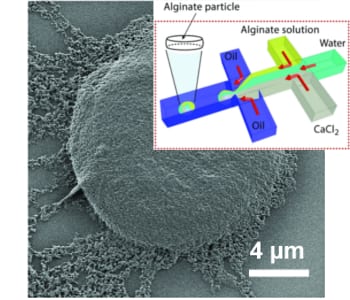
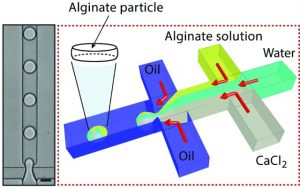
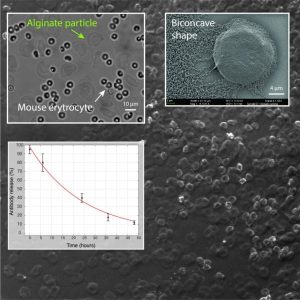 In their recent work, L. Mazutis and co-workers have developed a microfluidic approach for the production of alginate hydrogels having the unique biconcave shape and the size of a mammalian cell (~10 µm). Using a droplet microfluidics device with a characteristic flow-focusing junction that prevents premature alginate polymerization on-chip, authors have encapsulated fluorescently labeled IgG antibodies and followed their release in different buffers. Interestingly, the release of encapsulated antibodies was largely affected by the presence of phosphate ions that chelate calcium (a cross-linker of alginate polymer) and cause the dissolution of the hydrogel.
In their recent work, L. Mazutis and co-workers have developed a microfluidic approach for the production of alginate hydrogels having the unique biconcave shape and the size of a mammalian cell (~10 µm). Using a droplet microfluidics device with a characteristic flow-focusing junction that prevents premature alginate polymerization on-chip, authors have encapsulated fluorescently labeled IgG antibodies and followed their release in different buffers. Interestingly, the release of encapsulated antibodies was largely affected by the presence of phosphate ions that chelate calcium (a cross-linker of alginate polymer) and cause the dissolution of the hydrogel.
In addition, the authors tested the biocompatibility of their new alginate particles in whole blood. The results of this work should provide a better understanding of the conditions in which alginate particles could be used as drug delivery systems or biological implants. Finally, the microfluidics approach described in this work should be straightforward to adapt to other types of biopolymers and provide a better control over hydrogel particles size and shape.