Silver-coated microneedles used as probes for surface enhanced Raman scattering (SERS) measurements might enable future in vivo detection of intradermal components buried deeply under the skin.
 In the areas of immunology and diagnosis, intradermal component of human skin, such as dendritic cells and capillary blood are of interest. Several methods have been developed to probe intradermal layers. Among these methods, Raman spectroscopy is of special interest, because it promises to offer fingerprints related to structures, conformations, and processes in biomolecules. The challenge is that the relevant components are buried at 700 µm under the human skin or deeper – a depth which is difficult to probe by Raman spectroscopy. Moreover, the detection sensitivity is limited by the small Raman scattering cross section of endogenous biomolecules.
In the areas of immunology and diagnosis, intradermal component of human skin, such as dendritic cells and capillary blood are of interest. Several methods have been developed to probe intradermal layers. Among these methods, Raman spectroscopy is of special interest, because it promises to offer fingerprints related to structures, conformations, and processes in biomolecules. The challenge is that the relevant components are buried at 700 µm under the human skin or deeper – a depth which is difficult to probe by Raman spectroscopy. Moreover, the detection sensitivity is limited by the small Raman scattering cross section of endogenous biomolecules.
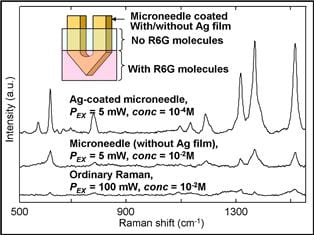
Surface-enhanced Raman scattering (SERS) techniques could be an alternative as Raman signals can be amplified up to 1014-fold by using metallic surfaces. However, the subcutaneous injection of metal nanoparticles is not practicable because they may be toxic. Scientists from the Nanyang Technological University (Singapore) now suggest the use of metal-coated microneedles.
For a proof of principle, they utilized a silver-coated microneedle-based SERS probe to examine two-layered phantoms that were comprised of elastic scatterers and absorbers to mimic the light absorption and scattering properties of human skin. The researchers were able to sensitively detect Rhodamine 6G and glucose test molecules buried deeper than 700 µm inside the mimics. They succeeded in quantifying glucose concentrations ranging from 5 to 150 mM. The study demonstrates the potential of the new microneedle probe for in vivo intradermal SERS measurements of analytes with clinical relevance.