Cold atmospheric plasmas (CAP) has been widely applied in the medical sector such as bacteria decontamination and wound healing given its unique multi‐modal characteristics. CAP has shown promising anti‐cancer efficacies over approximately 20 types of cancers in vitro as well. Researchers found that the plasma‐activated medium (PAM), obtained by treating the medium using plasma, can kill cancer cells as effectively as directly exposing cancer cells to CAP. Importantly, PAM outweighs direct CAP treatment by avoiding external physical and chemical influences such as ultraviolet, heat, electromagnetic field, dozens of reactive species, and can be persevered with activity in the refrigerator for at least 7 days that allows easy transportation of such a regime.
Breast cancer is a heterogeneous disease with increasing incidence and mortality worldwide. Triple‐negative (TN) breast cancers are known for their poor clinical outcome and lack of effective targeted therapy with no or mild adverse effect due to the absence of druggable surficial markers. PAM, as a physical approach with dose‐dependent cancer cell selectivity, represents a promising novel oncotherapeutic modality if properly controlled. The pivotal questions to be addressed before clinical application of PAM as an oncotherapy is how to define “dose” and quantitatively associate dose with the cancer cell killing effect: there is no quantitative assessment of the factors influencing its anti‐tumor efficacy and the mechanism driving its activity remains mysterious.
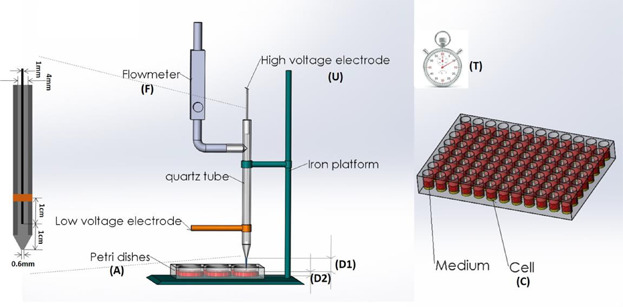
Schematic representation of the device and experimental setup
However, a team of Chinese researchers made progress through the use of 18 orthogonal experiments followed by linear model construction. In their study they identified four deterministic parameters, that is, “treatment time,” “liquid surface area,” “thickness of medium,” and “number of cells,” which play deterministic roles on the anti‐tumor efficacy of plasma. The team propose the “Reactive Species Diffusion Model” and “Signal Transduction Model” to explain the mechanics determining the activity of plasma treated medium and its cell‐death induction efficacy. They, in addition, propose the use of deterministic parameters in defining plasma dose in the liquid form to more objectively reflect its multi‐modality nature. This new study contributes in elucidating plasma activity and efficacy in a quantitative way, which guides other plasma related investigations and accelerates plasma clinical applications as a type of precision oncotherapy and a effective and safe treatment for breast cancer.











