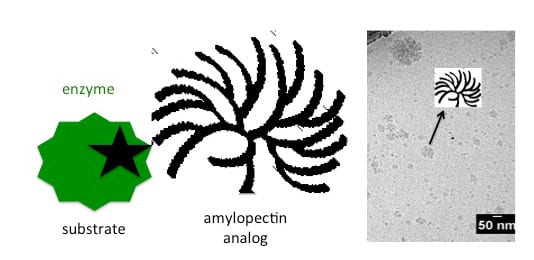
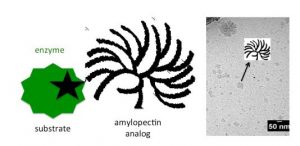
 Starch (amylose and amylopectin), glycogen and cellulose are natural polymers, and they are all around us. We eat them and use them on a daily basis but we do not understand them completely. Even though these sugars are simple concerning what they are made of (the repeating unit is glucose), they are hard to characterize. If there is a slight change in a structure, polymers’ properties can vary a lot. For instance, a molecule of amylose consists of many glucose units that are connected via α-(1,4) linkages creating a long linear chain, which is insoluble in water. Whereas amylopectin actually represents a long amylose chain with many shorter chains connected to it with α-(1,6) linkages, and is soluble in water. Or take a look at barely soluble-in-anything cellulose, which is also linear and consists of glucose units only connected via β-(1,4) linkages.
Starch (amylose and amylopectin), glycogen and cellulose are natural polymers, and they are all around us. We eat them and use them on a daily basis but we do not understand them completely. Even though these sugars are simple concerning what they are made of (the repeating unit is glucose), they are hard to characterize. If there is a slight change in a structure, polymers’ properties can vary a lot. For instance, a molecule of amylose consists of many glucose units that are connected via α-(1,4) linkages creating a long linear chain, which is insoluble in water. Whereas amylopectin actually represents a long amylose chain with many shorter chains connected to it with α-(1,6) linkages, and is soluble in water. Or take a look at barely soluble-in-anything cellulose, which is also linear and consists of glucose units only connected via β-(1,4) linkages.
In order to try to understand as much as possible about their structure and the relationship between molecular structure and the physical properties, the first steps are to create such polysaccharides. However, if you try to create these polymers via traditional organic chemistry, it is very hard and time consuming. Nevertheless if we copy nature and use enzyme as catalysts in the synthesis, the synthesis is very easy. Enzymes catalyze specific things, like formation of α-(1,4) or α-(1,6) linkages.
With this in mind, a team from the University of Groningen have published research which concentrates on the enzymatic synthesis of glycosidic polysaccharide analogs and their meticulous characterization. Gathered information are used for better characterization of natural polysaccharides. Their main objective was to outline different enzymatic routes to such polysaccharides and possibilities for characterization of both synthetic and natural ones.