 Zwitterion films with a significant bandwidth in the intermolecular band dispersion show evidence of electron pockets at the Fermi Level.
Zwitterion films with a significant bandwidth in the intermolecular band dispersion show evidence of electron pockets at the Fermi Level.
Most organic materials are insulators or semiconductors and few explicitly exhibit a density of states at the Fermi level. Stable charge neutral organic molecules do not usually behave as metals because the interatomic hybridization causes the conduction and valence bands to be completely unfilled and filled, respectively. This is to say that for most stable completely organic systems, the chemical potential resides well within the gap between the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO). There are exceptions, notably the tetrathiafulvalene-tetracyanoquinodimethane (TTF-TCNQ) system, and since 1973, over 400 organic conductors have been synthesized of which about 50 are superconducting, like the Bechgaard salts. Yet a single component organic conductor remains a Holy Grail of organic chemistry.
One of the general issues that touches on molecular conductors is the hybridization of molecular orbitals to form a well-defined band structure. Hybridization of molecular orbitals to form a band structure is fairly well established experimentally and theoretically. For larger molecules, an intramolecular band structure is far more likely and commonly observed. Intramolecular band dispersion has been documented in self-assembled monolayers, including polyphenyl species. In contrast, due to very small effective Brillouin zone (requiring exceptionally good wave vector resolution) and the generally much smaller intermolecular interactions, experimental evidence of band structure resulting from intermolecular interactions is seldom observed for ordered assemblies of large molecules (larger than ethylene, benzene and CO). Measuring an intermolecular band structure relevant to the conduction properties thus continues to remain a technical challenge for larger molecular species. In spite of the wide acceptance of band structure for molecular systems, when it comes to organic conductors, even for TTF-TCNQ related compounds and for Bechgaard salts, the density of states directly observed at the Fermi level is quite low.
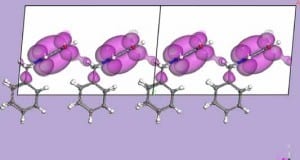
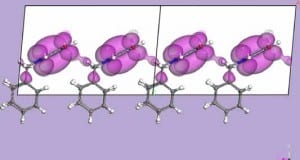
Now there is compelling evidence of electron pockets, at the Fermi Level, in the band structure for an organic zwitterion molecule of the p-benzoquinonemonoimine type. The electronic structure of the zwitterion molecular film has a definite, although small, density of states evident at the Fermi level as well as a nonzero inner potential and thus is very different from a true insulator. In spite of a small Brillouin zone, significant band width is observed in the intermolecular band dispersion. The results demonstrate that Bloch’s theorem applies to the wave vector dependence of the electronic band structure formed from the molecular orbitals of adjacent molecules in a molecular thin film of a p-benzoquinonemonoimine-type zwitterion. Further, the experimental and calculated band structure suggests that there is a significant density of molecular states at the Fermi level, related to the occurrence of electron pockets in thin films of (6Z)-4-(benzylamino)-6-(benzyliminio)-3-oxocyclohexa-1,4-dien-1-olate, a member of this family of benzoquinonemonoimine zwitterions (Figure 1, with R = CH2-C6H5). This is somewhat surprising because such large dipole molecules tend to be good dielectrics, but obviously molecular packing and orientation can enhance or suppress the dielectric properties.
The results show a distinct wave vector dependence of the electronic band structure formed from the molecular orbitals of adjacent molecules in a thin film of the zwitterion (6Z)-4-(benzylamino)-6-(benzyliminio)-3-oxocyclohexa-1,4-dien-1-olate deposited from solution on gold. Inter-molecular band dispersion has been previously observed for small adsorbates,but this zwitterion molecular system is a somewhat larger molecule that has a large intrinsic dipole and there is evidence of a finite density of states at the Fermi level.
This zwitterion molecular film is very different from a true insulator. The nonzero inner potential and the evidence of electron pockets, observed in the photoemission spectra, can be associated with electronic states which appear to be slightly filled or occupied in the solid state, but normally would be completely unoccupied in the single molecule. Our model calculations confirm that there is an electronic density shared inter-molecularly between the oxygen and nitrogen groups of the zwitterion crystal, consistent with experiment. One can therefore expect significant intrinsic conductivity of such films, making them of interest for organic electronics.