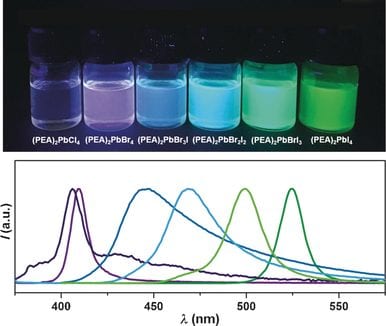
Since the isolation of graphene, a single-layer of graphite, in 2004, a large amount of research has been directed at preparing and isolating other 2D materials due to their unusual characteristics and wide applications. Organic-inorganic hybrid perovskites have been intensively studied, due to their excellent opto-electrical properties, such as high absorption coefficiency, strong photoluminescence and low trap-state density. Efforts to combine these appealing optical properties with the unique 2D structure is attracting increasing research interest. However, the synthesis of ultrathin 2D nanosheets (NSs) remains a significant challenge for perovskite materials.
In their recent communication published in Angew. Chem. Int. Ed., Hua Zhang and his colleagues at Nanyang Technological University in Singapore have demonstrated a facile crystallization method for the synthesis of free-standing 2D phenylethylammonium lead halide NSs. Specifically, based on the “like dissolves like” theory, perovskite precursors were first well dissolved in N,N-dimethylformamide, and the solution was then quickly dropped into a poor solvent (e.g., toluene) under vigorous stirring. Accompanied by an immediate color change of the solution, perovskite NSs can be obtained through this simple method. The effect of solvents is crucial to the crystallization process and was systematically investigated. It was found that the high polarity solvent could provide higher saturation concentration of perovskite precursors, and thus favoring the formation of smaller sized perovskite NSs.
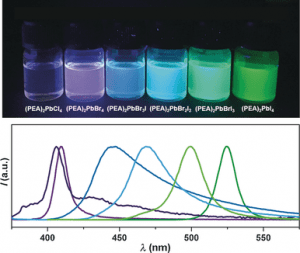 The as-prepared perovskite NSs exhibit much better stability under ambient conditions and light irradiation compared to their conventional 3D counterparts (e.g., CH3NH3PbI3 perovskite quantum dots), which might have potential applications in solar cells, light-emitting diodes, photodetectors, and field-effect transistors.
The as-prepared perovskite NSs exhibit much better stability under ambient conditions and light irradiation compared to their conventional 3D counterparts (e.g., CH3NH3PbI3 perovskite quantum dots), which might have potential applications in solar cells, light-emitting diodes, photodetectors, and field-effect transistors.