Short chain polyacetals and their corresponding copolymers have found applications in fields ranging from medical devices to electronic compounds. In contrast, long-chain polyacetals have found little attention, probably due to a lack of conveniently accessible and sufficiently pure building blocks. Given their long methylene sequences plant oils are attractive candidates for this purpose.
Short chain polyacetals and their corresponding copolymers have found applications in fields ranging from medical devices to electronic compounds. In contrast, long-chain polyacetals have found little attention, probably due to a lack of conveniently accessible and sufficiently pure building blocks. Given their long methylene sequences plant oils are attractive candidates for this purpose.
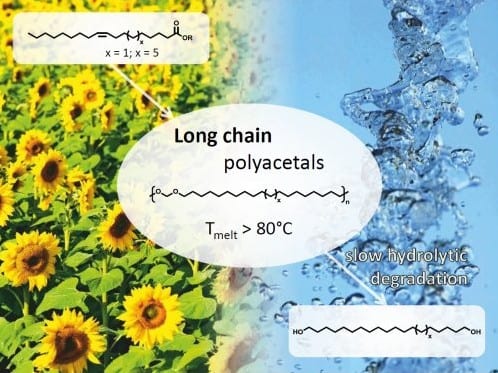
The group of Stefan Mecking (University of Konstanz, Germany) now shows how the polycondensation of plant oil-derived diols provides access to novel long-chain polyacetals. These materials exhibit, in comparison to mid-chain polyacetal, significantly higher melting and crystallization temperatures, which are at the lower end of typical thermoplastic materials. Typical for polyacetals, the novel materials are hydrolytically degradable. The degradation rates observed for the long-chain polyacetals are clearly lower compared with mid-chain length polyacetal. This might suggest that the degradation rates of such polyacetals, in general, can be designed over a wide range via the methylene sequence length.
The accessibility from renewable resources as well as the interesting hydrolytic degradation behavior of these polyacetals render these results an important contribution to the field of renewable materials.