Stimuli-responsive hydrogels have attracted increasing interest due to their potential application in the biomedical field, for example as drug delivery carriers. A highly efficient and biocompatible trigger for release from hydrogels is a change in reduction-potential which is a unique and ubiquitous signal in cancer cells and tumor tissue and can lead to a rapid and efficient drug release. Therefore, the design of hydrogel systems with easy production and fast response to reducing stimuli are an active area of research.
1,4-dithio-DL-threitol (DTT), glutathione tripeptide (γ-glutamyl-cysteinyl-glycine; GSH) and tris(2-carboxyethyl) phosphine (TCEP) are commonly used as disulfide-cleaving agents in a biological context. As a cellular reducing agent,the concentration of GSH (2-10 mM) in the cytosol is 100-1000 times higher than that in the extracellular fluids and circulation (about 2-20 μM). In tumor cells the GSH concentration is at least four times higher compared to normal tissues and disulfide bonds of a nano-vehicles can be cleaved, leading to the disassembly of the vehicle and the subsequently release of an anticancer drug.
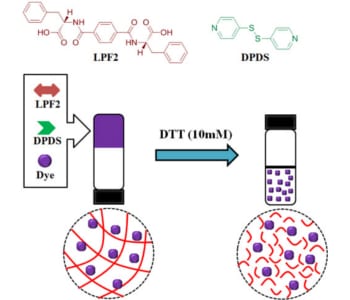
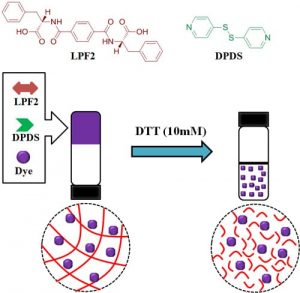 At Shanghai Jiao Tong University, Prof. Chuanliang Feng and co-workers are working on novel stimulus-responsive hydrogels self-assembled from low-molecular-weight gelators and their applications in controlling cell adhesion. Recently, they have successfully explored a novel and easy approach to construct biocompatible, redox-responsive supramolecular hydrogels co-assembled from two small organic molecules to encapsulate dyes. Furthermore, the disulfide-containing nanofibrous hydrogel is able to control the release of encapsulated dyes in response to reductive conditions mimicking the intracellular environment of tumor tissue.
At Shanghai Jiao Tong University, Prof. Chuanliang Feng and co-workers are working on novel stimulus-responsive hydrogels self-assembled from low-molecular-weight gelators and their applications in controlling cell adhesion. Recently, they have successfully explored a novel and easy approach to construct biocompatible, redox-responsive supramolecular hydrogels co-assembled from two small organic molecules to encapsulate dyes. Furthermore, the disulfide-containing nanofibrous hydrogel is able to control the release of encapsulated dyes in response to reductive conditions mimicking the intracellular environment of tumor tissue.
This work provides a novel and simple strategy for preparing redox-responsive supramolecular hydrogel, which may pave a new way to easily fabricated stimulus-response nanomaterials.