The immunological function of nanomaterials is one of the hottest research areas in material science. However, many reports indicate that efficient immune response is always generated by biomacromolecules, such as proteins and polypeptides, not simple nanoparticles without any functional modification.
Recently, Prof. Chunying Chen and her colleagues, from National Center for Nanoscience and Technology, China, demonstrated that polyhydroxylated metallofullerenol, in particular gadolinium endohedral metallofullerenol (Gd@C82(OH)22), can significantly activate primary mouse macrophages to produce pro-inflammatory cytokines IL-1b, without a second stimulus. IL-1β is a well-known cytokine, secreted by macrophages, which initiates inflammatory responses to clear bacterial infection, recruit neutrophils, and co-stimulate T cells. Recently, it has been proved that the action of IL-1b on specific CD8+ T cells may contribute to therapeutically relevant anticancer immune responses.
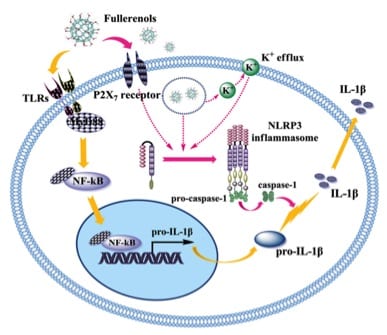
 In macrophages, secretion of IL-1b is always related to two indispensable and sequential pathways, a “priming” pathway, TLRs/MyD88/NF-kB pathway (yellow arrows), to generate pro-IL-1b genes,and a second pathway via NLRP3 inflammasome activation(red arrows), and caspase-1 activation (red arrows) to promote the maturation of bioactive IL-1b. As demonstrated in the figure, activation of TLR would stimulate expression of NF-kB, which was crucial for the production of pro-IL-1b. Maturation of pro-IL-1b required a signal from caspase-1. Meanwhile NLRP3 inflammasome was crucial for the activation of caspase-1. In previous works, nanoparticles always could not stimulate the two different paths and generate IL-1b by itself. A second stimulus, e.g. LPS is necessary. However, in this recently published paper, Gd@C82(OH)22 was proved to individually and efficiently initiate these two pathways, both in vitro and in vivo, resulting high expression of IL-1b, which definitely will shed light on antitumor chemotherapies. I believe that this work may open a new gate for chemical therapy of cancer by using nanoparticles and future work from Chen’s group to unveil the mechanism of this rare dual stimulation activity is expected.
In macrophages, secretion of IL-1b is always related to two indispensable and sequential pathways, a “priming” pathway, TLRs/MyD88/NF-kB pathway (yellow arrows), to generate pro-IL-1b genes,and a second pathway via NLRP3 inflammasome activation(red arrows), and caspase-1 activation (red arrows) to promote the maturation of bioactive IL-1b. As demonstrated in the figure, activation of TLR would stimulate expression of NF-kB, which was crucial for the production of pro-IL-1b. Maturation of pro-IL-1b required a signal from caspase-1. Meanwhile NLRP3 inflammasome was crucial for the activation of caspase-1. In previous works, nanoparticles always could not stimulate the two different paths and generate IL-1b by itself. A second stimulus, e.g. LPS is necessary. However, in this recently published paper, Gd@C82(OH)22 was proved to individually and efficiently initiate these two pathways, both in vitro and in vivo, resulting high expression of IL-1b, which definitely will shed light on antitumor chemotherapies. I believe that this work may open a new gate for chemical therapy of cancer by using nanoparticles and future work from Chen’s group to unveil the mechanism of this rare dual stimulation activity is expected.
Abbreviations: IL-1b: Interleukin-1b; CD8: Cluster of Differentiation 8; TLR: Toll-like Receptor; MyD88: Myeloid Differentiation factor 88; NF-kB: Nuclear Factor-kappa B; NLRP3: Nod-like receptor (NLR)-family protein 3; pro-IL-1b: Interleukin 1 beta precursor; LPS: Lipopolysaccharide