 Dithiolethiones, are a well-known class of chemo-preventive and antioxidant agents. But despite their potential in cancer therapy and prevention, clinical trials of dithiolethiones have resulted in questionable efficacy as well as significant side effects. This may be due to the unfavorable properties of these drugs such as insufficient accumulation in tumor tissue, cytotoxicity to normal cells, quick removal from the blood by the kidneys and low aqueous solubility.
Dithiolethiones, are a well-known class of chemo-preventive and antioxidant agents. But despite their potential in cancer therapy and prevention, clinical trials of dithiolethiones have resulted in questionable efficacy as well as significant side effects. This may be due to the unfavorable properties of these drugs such as insufficient accumulation in tumor tissue, cytotoxicity to normal cells, quick removal from the blood by the kidneys and low aqueous solubility.
In recent years, polymeric nanoparticles have attracted growing attention as drug carriers to improve therapeutic efficacy and minimize side effects. Nanoparticles have been shown to prolong systemic circulation time, enable site-specific delivery of drugs, and solubilize and stabilize drug molecules.
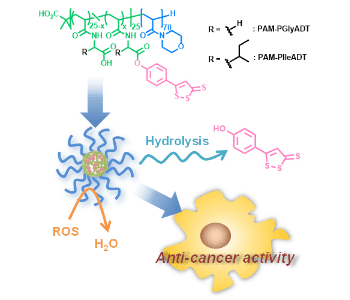
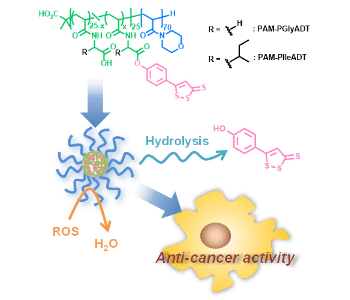
In their recent work, Hasegawa et al. developed polymeric prodrug micelles for delivery of the dithiolethione drug 5-(4-hydroxyphenyl)-3H-1,2-dithiole-3-thione (ADT-OH). They designed and synthesized amphiphilic diblock copolymers composed of a hydrophilic segment and a hydrophobic segment linking ADT-OH via an ester bond using two different linkers.
When dispersed in water, the resulting amphiphilic polymers self-assembled to form micellar nanostructures of about 40-50 nm in diameter. By using different linkers, the authors were able to control the release of ADT-OH from the micelles under physiological conditions. Furthermore, the micelles could neutralize hydrogen peroxide, one of reactive oxygen species (ROS) that is known to be produced in cancer cells and stimulate tumor growth and metastasis. The micelles were capable of killing human breast, colon and liver cancer cells. This micellar approach could potentially lead to new ways of using dithiolethione drugs in cancer therapy and prevention.