To fulfill the complex requirements of medical applications, multi-functional biomaterials need to be designed. Shape-memory, stimuli-sensitivity, degradability, and controlled drug release are important functions for biomedical applications and often a combination of these functions is required. Together, Prof. A. Lendlein and Prof. A. Pandit have guest-edited a special issue on Advanced Functional Polymers Addressing the Needs of Modern Medicine in Macromolecular Rapid Communications dealing with a variety of highly functional polymers and their application towards medical challenges.
To fulfill the complex requirements of medical applications, multi-functional biomaterials need to be designed. Shape-memory, stimuli-sensitivity, degradability, and controlled drug release are important functions for biomedical applications and often a combination of these functions is required. Together, Prof. A. Lendlein and Prof. A. Pandit have guest-edited a special issue on Advanced Functional Polymers Addressing the Needs of Modern Medicine in Macromolecular Rapid Communications dealing with a variety of highly functional polymers and their application towards medical challenges.
As the contributions to this special issue show, the design of functional polymers for medicine is a broad research field. To provide you with an insight into this exciting and interdisciplinary field the following 3 articles will be available for free for a limited time.
A Combinatorial Approach for Porous Polymer Structures
 A novel method for the simultaneous preparation of a large number of porous polymeric structures with highly differing physical properties was developed by Prof. D. W. Grijpma and co-workers. Low molecular weight methacrylate end-functionalized polymers (macromers) were dissolved in ethylene carbonate, cooled below the melting temperature of the solvent, and subsequently photocross-linked. The crystallized and phase-separated ethylene carbonate could be extracted with water, upon which a porous crosslinked polymer network was obtained. The method was applied to combinatorial mixtures of methacrylate end-functionalized polymers that are relevant in the biomedical field: poly(trimethylene carbonate-dimethacrylate), poly(D,L-lactide-dimethacrylate), and poly(ethylene glycol-dimethacrylate) dissolved in ethylene carbonate at concentrations of approximately 25 wt%. In this manner, 63 different porous polymeric structures with a very wide range of physical properties were prepared simultaneously. In the hydrated state the compressive moduli of the prepared structures ranged from 0.01 to 60 MPa, as water uptake ranged between 3 and 1500 wt%.
A novel method for the simultaneous preparation of a large number of porous polymeric structures with highly differing physical properties was developed by Prof. D. W. Grijpma and co-workers. Low molecular weight methacrylate end-functionalized polymers (macromers) were dissolved in ethylene carbonate, cooled below the melting temperature of the solvent, and subsequently photocross-linked. The crystallized and phase-separated ethylene carbonate could be extracted with water, upon which a porous crosslinked polymer network was obtained. The method was applied to combinatorial mixtures of methacrylate end-functionalized polymers that are relevant in the biomedical field: poly(trimethylene carbonate-dimethacrylate), poly(D,L-lactide-dimethacrylate), and poly(ethylene glycol-dimethacrylate) dissolved in ethylene carbonate at concentrations of approximately 25 wt%. In this manner, 63 different porous polymeric structures with a very wide range of physical properties were prepared simultaneously. In the hydrated state the compressive moduli of the prepared structures ranged from 0.01 to 60 MPa, as water uptake ranged between 3 and 1500 wt%.
Electrical Control of Cell Differentiation
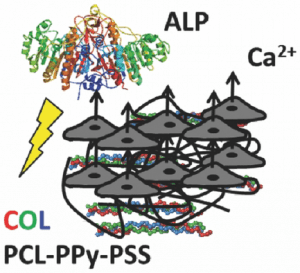 In tissue engineering, proregenerative biomaterials for the treatment of bone conditions and disorders that require surgical intervention are of growing importance in modern societies in which life expectancy are increasing. Bone tissue scaffolds that instruct the behavior of cells residing within them are particularly interesting for such applications.
In tissue engineering, proregenerative biomaterials for the treatment of bone conditions and disorders that require surgical intervention are of growing importance in modern societies in which life expectancy are increasing. Bone tissue scaffolds that instruct the behavior of cells residing within them are particularly interesting for such applications.
Prof. C. E. Schmidt and co-workers describe the preparation of conductive fiber–based bone tissue scaffolds (nonwoven mats of electrospun polycaprolactone with an interpenetrating network of polypyrrole and polystyrenesulfonate) that enable the electrical stimulation of human mesenchymal stem cells to enhance their differentiation toward osteogenic outcomes. Importantly, electrical stimulation increased alkaline phosphatase activity and calcium deposition, which represents an important step toward the formation of calcified bone-like extracellular matrix; such materials should be relatively non-immunogenic in vivo and represent interesting platforms for further development of conductive tissue scaffolds.
Proteins make Gold Nanoparticles
 The use of biomolecules to synthesize inorganic nanomaterials, including metallic nanoparticles, offers the ability to induce controlled growth under mild environmental conditions. Prof. A. Mizeres and co-workers used the recently discovered silk-like “suckerin” proteins to induce the formation of gold nanoparticles (AuNPs). Advantage was taken of the distinctive biological and physico-chemical characteristics of suckerins, namely their facile recombinant expression, their solubility in aqueous solutions, and their modular primary structure with high molar content of redox-active tyrosine residues to induce the formation of AuNPs not only in solution, but also from nano-structured solid substrates like nanotubes fabricated from suckerins. This straightforward coating method could be extended to a wider range of morphologies and could find applications in solid-state biosensors.
The use of biomolecules to synthesize inorganic nanomaterials, including metallic nanoparticles, offers the ability to induce controlled growth under mild environmental conditions. Prof. A. Mizeres and co-workers used the recently discovered silk-like “suckerin” proteins to induce the formation of gold nanoparticles (AuNPs). Advantage was taken of the distinctive biological and physico-chemical characteristics of suckerins, namely their facile recombinant expression, their solubility in aqueous solutions, and their modular primary structure with high molar content of redox-active tyrosine residues to induce the formation of AuNPs not only in solution, but also from nano-structured solid substrates like nanotubes fabricated from suckerins. This straightforward coating method could be extended to a wider range of morphologies and could find applications in solid-state biosensors.